|
|
| (20 intermediate revisions not shown) |
| Line 1: |
Line 1: |
| - | <center>[[Davidson Missouri W| <span style="color:black">Home</span>]] | [[Davidson Missouri W/Background Information| <span style="color:red">Background Information</span>]] | [[Davidson Missouri W/Solving the HPP in vivo| <span style="color:red">Current Project: Solving the Hamiltonian Path Problem ''in vivo''</span>]] | [[Davidson Missouri W/Mathematical Modeling| <span style="color:red">Mathematical Modeling</span>]] | [[Davidson Missouri W/Gene splitting| <span style="color:red"> Gene Splitting </span>]] | [[Davidson Missouri W/Controlling Expression| <span style="color:red"> Controlling Expression </span>]] | [[Davidson Missouri W/Traveling Salesperson Problem| <span style="color:red">Traveling Salesperson Problem</span> ]] | [[Davidson Missouri W/Software|<span style="color:red">Software</span>]] | [[Davidson Missouri W/Resources and Citations|<span style="color:red">Resources and Citations</span>]]</center> | + | <center>[[Davidson Missouri W| <span style="color:red">Home</span>]] | [[Davidson Missouri W/Background Information| <span style="color:red">Background Information</span>]] | [[Davidson Missouri W/Solving the HPP in vivo| <span style="color:red">Current Project: Solving the Hamiltonian Path Problem ''in vivo''</span>]] | [[Davidson Missouri W/Mathematical Modeling| <span style="color:red">Mathematical Modeling</span>]] | [[Davidson Missouri W/Gene splitting| <span style="color:black"> Gene Splitting </span>]] | [[Davidson Missouri W/Results| <span style="color:red">Results</span>]] | [[Davidson Missouri W/Traveling Salesperson Problem| <span style="color:red">Traveling Salesperson Problem</span> ]] | [[Davidson Missouri W/Software|<span style="color:red">Software</span>]] | [[Davidson Missouri W/Resources and Citations|<span style="color:red">Resources and Citations</span>]]</center> |
| | <hr> | | <hr> |
| | | | |
| - | We have selected 4 genes to split. We will use our [http://gcat.davidson.edu/iGEM07/genesplitter.html online] gene splitting [[Davidson Missouri W/Web tool| web tool]] to choose the PCR primers. Davidson will produce these 4 split genes and test each one. | + | =What is Gene Splitting?= |
| | + | |
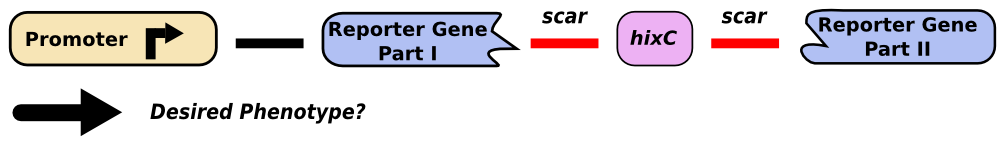
| | + | "Gene splitting" refers to the insertion of a ''hixC'' site within the coding region of a gene. Although this allows us to create edges for the simulation of a graph, it will change the protein sequence, potentially interfering with proper functionality. We successfully inserted ''hixC'' in two different reporter genes, GFP and RFP. Cells transformed with plasmids containing these "split" genes still fluoresce the appropriate color. |
| | + | |
| | + | [[Image:Unsplit-gene.png|thumb|440px|none|A promoter upstream of a reporter gene will produce the expected phenotype.]] |
| | + | |
| | + | [[Image:Split-gene.png|thumb|830px|none|The insertion of a ''hixC'' site, or 13 extra amino acids, might prevent the expected phenotype.]] |
| | + | |
| | + | To facilitate the splitting process we developed software to help us. Our [http://gcat.davidson.edu/iGEM07/genesplitter.html online] gene splitting web tool (click [[Davidson Missouri W/Web tool| here]] for a tutorial) helps us choose PCR primers that will amplify the appropriate segments of a gene of interest. |
| | | | |
| - | # [[Davidson Missouri W/Gene splitting#Kanamycin|Kanamycin Nucleotidyltransferase]]
| |
| - | # [[Davidson Missouri W/Gene splitting#RFP|Red Fluorescent Protein]]
| |
| - | # [[Davidson Missouri W/Gene splitting#CAT|Chloramphenicol Acetyltransferase]]
| |
| - | # [[Davidson Missouri W/Gene splitting#Cre|Cre Recombinase]]
| |
| - | <br>
| |
| - | [[Davidson Missouri W|Return to DMW main page]]
| |
| - | <br>
| |
| | =The Genes= | | =The Genes= |
| | + | |
| | {| border="1" cellpadding="5" cellspacing="0" align="center" width="100%" | | {| border="1" cellpadding="5" cellspacing="0" align="center" width="100%" |
| | |- | | |- |
| | !style="color: red; background-color: black;" width="5%"| Gene | | !style="color: red; background-color: black;" width="5%"| Gene |
| | !style="color: red; background-color: black;" width="65%"| Description | | !style="color: red; background-color: black;" width="65%"| Description |
| - | !style="color: red; background-color: black;" width="30%"| Graphic | + | !style="color: red; background-color: black;" width="30%"| Protein 3D Structure and ''hixC'' Insertion Point |
| | |- | | |- |
| | | | |
| - | |style="color: black; background-color: red;" align="center"|Kanamycin Nucleotidyltransferase | + | |style="color: black; background-color: green;" align="center"|GFP - Green Fluorescent Protein |
| - | |
| + | |
| - | <span id="Kanamycin"></span>One gene our team will be using as a node in our Hamiltonian Path problem is Kanamycin resistance translated in the form of Kanamycin nucleotidyltransferase (KNTase). The antibiotic Kanamycin, once in the cytosol of E.Coli, inhibits protein synthesis by interacting with the “decoding” region in the small ribosomal subunit RNA.(Sambrook and Russel, 2001) The KNTase enzyme, as a member of the aminoglycoside phosphotransferase (APH) enzyme family, blocks Kanamycin’s ability to inhibit protein synthesis by transferring a nucleoside monophosphate (adenyl) group from Mg2+-ATP to the 4’ hydroxyl group of Kanamycin, inhibiting its ability to bind to the srRNA.
| + | |
| - | [http://www.ingentaconnect.com/content/els/00452068/1999/00000027/00000005/art91144 1]
| + | |
| | | | |
| - | Our goal was to insert a hix site (a polar molecule) in an area of KNTase protein that would not interfere with its ability to inhibit Kanamycin. We looked at mutational analysis of KNTase and other aminoglycoside phosphotransferase enzymes to determine which aspects of KNTase’s structure were integral to its function and therefore not an ideal site for hix site insertion. KNTase is a dimer consisting of 253 amino acids in the molecule [http://www.bioscience.org/1999/v4/d/wright/fulltext.htm 3]. In looking at conserved structures in the APH family we took into consideration that:
| + | |In the spring of 2007, Karen Acker pioneered the gene-splitting process by splitting GFP. According to previous research, it had been shown that it was possible to insert extra amino acids within a particular region of the protein without disrupting green fluorescence. Acker successfully inserted a ''hixC'' site at the same location, between amino acids 157 and 158. |
| | | | |
| - | -Substitution of AA 190 caused 650-fold decrease in enzyme activity [http://www.bioscience.org/1999/v4/d/perlin/fulltext.htm 2]
| + | |align="center"|[[Image:Gfp_insertion.png]] |
| | | | |
| - | -AA 190 is involved in catalysis [http://www.bioscience.org/1999/v4/d/wright/fulltext.htm 5]
| |
| - |
| |
| - | -AA 195 and 208 are involved in Mg2+ binding [http://www.bioscience.org/1999/v4/d/wright/fulltext.htmv 5]
| |
| - |
| |
| - | -Mutant Enzymes 190, 205, 210 all showed changes in mg+2 binding from the WT [http://www.bioscience.org/1999/v4/d/perlin/fulltext.htm 2]
| |
| - |
| |
| - | -Substitution of AA 210 (conserved) reduced enzyme activity [http://www.bioscience.org/1999/v4/d/perlin/fulltext.htm 2]
| |
| - |
| |
| - | -AA 166 serves to catalyze reactions involving ATP [http://www.bioscience.org/1999/v4/d/perlin/fulltext.htm 2]
| |
| - |
| |
| - | -AA 44 is involved in ATP binding [http://www.bioscience.org/1999/v4/d/wright/fulltext.htm 5]
| |
| - |
| |
| - | -AA 60 is involved in orientation of AA 44 and ATP binding [http://www.bioscience.org/1999/v4/d/wright/fulltext.htm 5]
| |
| - |
| |
| - | -We did not consider any Amino Acids near the N or C terminus
| |
| - |
| |
| - | -We did not consider any residues near ß-sheets or ∂-helices close to the active site because hydrogen bonding plays an active role in substrate stabilization and the polarity of our hix site could disrupt the secondary structure and therefore the hydrogen bonding ability of KNTase)
| |
| - |
| |
| - | |align="center"| [[Image:KNTase_hix_cut.png]] <br>The yellow bands at the top and bottom of the molecule denotes hix site insertion
| |
| - |
| |
| - | We decided to insert our hix sites at the 125 AA of each monomer due to their distance from each other, active site secondary structure, N or C terminus, and lack of any previous mutational analysis proving its function as integral.
| |
| | |- | | |- |
| | | | |
| | |style="color: black; background-color: red;" align="center"|DsRed - Red Fluorescent Protein | | |style="color: black; background-color: red;" align="center"|DsRed - Red Fluorescent Protein |
| | | | |
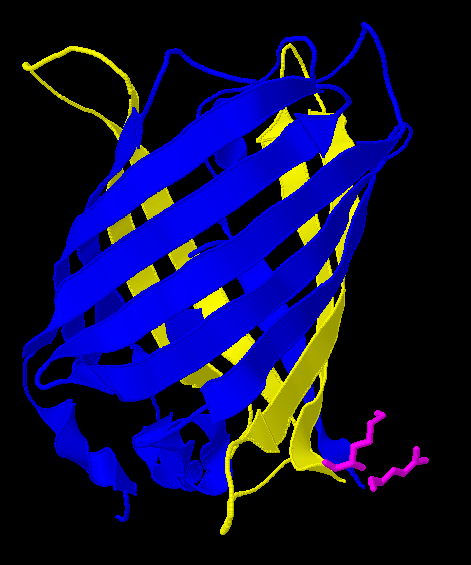
| - | | | + | |After Acker demonstrated that GFP fluoresces despite a ''hixC'' insertion, we tested the same process on RFP, [http://partsregistry.org/Part:BBa_E1010 RFP] (from [http://www.ncbi.nlm.nih.gov/Taxonomy/Browser/wwwtax.cgi?id=86600 ''Discosoma sp.'']). Although its DNA sequence is markedly different from GFP's, it has some amino acid similarity and a remarkable structural similarity. Both proteins have a Beta-barrel structure which surrounds an internal chromophore. |
| - | <span id="RFP"></span>We use genes to represent the nodes on our Hamiltonian path. One of the essential features of these genes is that they can tolerate the insertion of a Hix site. It has been previously demonstrated that GFP fluoresces despite a Hix insertion. Another glowing protein, [http://partsregistry.org/Part:BBa_E1010 RFP] (from [http://www.ncbi.nlm.nih.gov/Taxonomy/Browser/wwwtax.cgi?id=86600 ''Discosoma sp.'']), is a candidate for use in our path. Although its DNA sequence is markedly different from GFP's, it has some amino acid similarity and a remarkable structural similarity. Both proteins have a Beta-barrel structure which surrounds an internal chromophore.
| + | |
| | | | |
| - | Inserting 13 amino acids into a protein can potentially disrupt its ability to function. It is thus essential to find an insertion point that does not interfere with the protein's function. Fortunately, the similarity between GFP and RFP allows us to make a highly educated guess for where to insert. RFP's amino acid position 154 is homologous to GFP's amino acid position 157, which is where GFP was split. This is therefore our best guess for where to insert the Hix site.
| + | Fortunately, the similarity between GFP and RFP allowed us to make an educated guess for where to insert. RFP's amino acid position 154 is homologous to GFP's amino acid position 157, which is where GFP was split. This was therefore our best guess for where to insert the ''hixC'' site. |
| | | | |
| - | |align="center"|[[Image:Rfp_hix_insertion_point.jpg|200px]]
| + | RFP was also successfully split. However, the color is much weaker and takes a day at room temperature to become visible by eye. |
| - | |-
| + | |
| | | | |
| - | |style="color: black; background-color: red;" align="center"|Chloramphenicol Acetyltransferase | + | |align="center"|[[Image:Rfp_insertion.png]] |
| - | |<span id="CAT"></span>[http://partsregistry.org/Part:BBa_P1004 Chloramphenicol Acetyltransferase] was one of the genes we chose as a node for our Hamiltonian Path. It is a bacterial gene that neutralizes the effect of an antibiotic, Chloramphenicol, by transferring acetyl groups to Chloramphenicol and changes its shape into a harmless form.
| + | |
| | | | |
| - | The specific Chloramphenicol Acetyltransferase gene we are using comes from the plasmid PSV2CAT whose original source is an ''E. coli'' transposable element Tn9 (Sambrook, 2001) Its PDB ID# is [http://www.pdb.org/pdb/explore/explore.do?structureId=1PD5 1PD5].
| + | |} |
| | | | |
| - | I have chosen to insert my hixC site between amino acid 52 and 53. I chose this point because it is away from the active site of the protein, the point that contains the catalytic binding sites and allow the recognition and binding of the substrate. It is important for the insertion point to be away from the active site because we do not want the overall function and structure of the protein to be destroyed in the process of splitting. We want to split at a point where the two halves of the protein cannot work as single units, but once a hixC site has been inserted, and the two halves are brought back together, the protein displays its original function.
| + | =A Closer Look= |
| | | | |
| - | |align="center"|[[Image: Chlor.jpg|200px]]<br>The structure of a type I Chloramphenicol Acetyltransferase used in the BioBrick Registry.
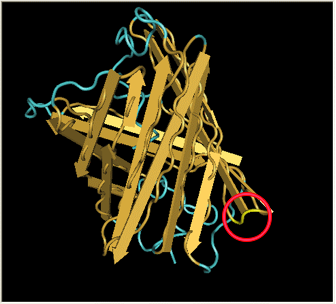
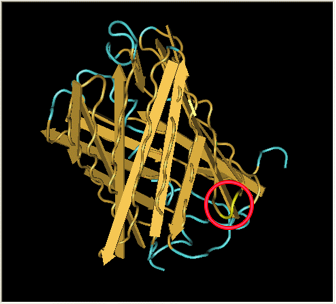
| + | Here is a more detailed view of splitting GFP. The image on the left, with the protein structure highlighted in green, shows the normal, wild-type structure of the protein. The image on the right highlights the two parts of the protein on either side of the ''hixC'' insertion, shown in pink. The additional 13 amino acids are not shown, but they would extend from one pink amino acid into the next. |
| - | |-
| + | |
| | | | |
| - | |style="color: black; background-color: red;" align="center"|Cre Recombinase
| + | From these 3-dimensional PDB images it becomes apparent that the insertion likely sticks into open space away from the rest of the protein. This increases the likelihood that the addition does not interfere with the beta barrel structure or with the chromophore. |
| - | |<span id="Cre"></span>We will also attempt to insert a hixC site into the Cre Recombinase gene. Cre Recombinase binds as a dimer to a specific sequence of DNA called a ''loxP'' site. If two ''loxP'' sites are facing in opposite orientations, then Cre Recombinase will flip the section of DNA in between. If two ''loxP'' sites are facing in the same orientation, then Cre Recombinase will excise the DNA in between, creating a new plasmid.
| + | |
| | | | |
| - | In researching Cre Recombinase, we found that the gene had already been split by another lab. [http://www3.interscience.wiley.com/cgi-bin/abstract/104558885/ABSTRACT] In the study done by Casanova et al, two independent but overlapping sections of the Cre Recombinase gene were placed in separate locations along an E. Coli chromosome. When translated, the two overlapping halves of the Cre Recombinase protein bound together and formed a functional Cre Recombinase protein.
| + | As a consequence of RFP's similar 3-dimensional structure it is assumed the ''hixC'' insertion acts in a similar fashion. |
| | | | |
| - | In order for Casanova et al's split protein to be functional, the overlapping section of the bound protein at the split site presumably could not significantly hinder the protein’s ability to bind to ''loxP'' sites or recombine DNA segments. With that in mind, we investigated the same region split by Casanova et al. as the prime candidate for the insertion of a hixC site.
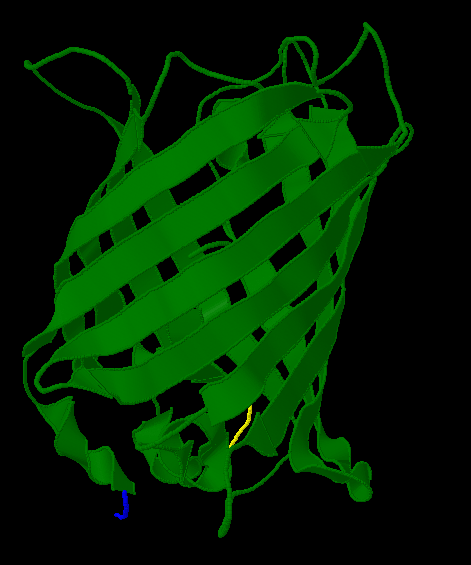
| + | [[Image:Gfp-nosplit.png|thumb|340px|left|The unmodified structure of GFP.]] |
| | + | [[Image:Gfp-split.png|thumb|340px|none|The sections before and after the ''hixC'' insertion are highlighted in blue and yellow. The amino acids at the insertion point are shown in pink.]] |
| | | | |
| - | The site that was eventually chosen reflects both the protein structure shown to the right and the previous research done in the Casanova lab. We believe that amino acids 190-191 along the Cre Recombinase protein are unlikely to play a significant role in the functioning of the protein, thus we decided on this location for the insertion of our hixC site.
| + | <br> |
| - | | + | <hr> |
| - | Figure 1 on the right depicts a monomer of Cre Recombinase bound to a DNA strand that it is about the cut. Our split site is highlighted in yellow and can be seen far away from the active site of the molecule.
| + | <center> |
| - | | + | <[[Davidson Missouri W/Mathematical Modeling | Previous Section]] | [[Davidson Missouri W/Results | Next Section>]] |
| - | Figure 2 on the right depicts two dimers of Cre Recombinase coming come together to cut DNA at two ''loxP'' sites. The site of our hixC insertion is highlighted in yellow on each molecule and can be seen far away from the active site.
| + | </center> |
| - | | + | |
| - | |align="center"|[[Image:Cre_recombinase_monomer1.png|200px]]<br>
| + | |
| - | Figure 1
| + | |
| - | | + | |
| - | [[Image:Cre_recombinase_tetramer1.png|200px]]<br> | + | |
| - | Figure 2
| + | |
To facilitate the splitting process we developed software to help us. Our [http://gcat.davidson.edu/iGEM07/genesplitter.html online] gene splitting web tool (click here for a tutorial) helps us choose PCR primers that will amplify the appropriate segments of a gene of interest.
Here is a more detailed view of splitting GFP. The image on the left, with the protein structure highlighted in green, shows the normal, wild-type structure of the protein. The image on the right highlights the two parts of the protein on either side of the hixC insertion, shown in pink. The additional 13 amino acids are not shown, but they would extend from one pink amino acid into the next.
From these 3-dimensional PDB images it becomes apparent that the insertion likely sticks into open space away from the rest of the protein. This increases the likelihood that the addition does not interfere with the beta barrel structure or with the chromophore.