BerkiGEM2007Present5
From 2007.igem.org
(→Phenotype of dead cells) |
|||
| Line 4: | Line 4: | ||
---- | ---- | ||
| + | |||
| + | '''Introduction''' <br> | ||
| + | To prevent chance of infection or unwanted proliferation after hemoglobin production, we have engineered a genetic self-destruct mechanism whereby when induced, the bacterial cell will express a genetic material-degrading toxin which kills the cell, but leaves it physically intact. <br> | ||
Growth control in our system is established by the incorporation of a plasmid that can be triggered to translate a toxin. The toxins are endonucleases or rnases that destroy the genetic material within a bacteria and thus prevent the bacteria from replicating. Throughout the summer, we worked on several different constructs of an inducible toxin and selected for the ones with the best phenotype. | Growth control in our system is established by the incorporation of a plasmid that can be triggered to translate a toxin. The toxins are endonucleases or rnases that destroy the genetic material within a bacteria and thus prevent the bacteria from replicating. Throughout the summer, we worked on several different constructs of an inducible toxin and selected for the ones with the best phenotype. | ||
| Line 9: | Line 12: | ||
---- | ---- | ||
| - | |||
| - | |||
Revision as of 04:26, 24 October 2007
<< Back to UC Berkeley iGEM 2007
Growth Control
Introduction
To prevent chance of infection or unwanted proliferation after hemoglobin production, we have engineered a genetic self-destruct mechanism whereby when induced, the bacterial cell will express a genetic material-degrading toxin which kills the cell, but leaves it physically intact.
Growth control in our system is established by the incorporation of a plasmid that can be triggered to translate a toxin. The toxins are endonucleases or rnases that destroy the genetic material within a bacteria and thus prevent the bacteria from replicating. Throughout the summer, we worked on several different constructs of an inducible toxin and selected for the ones with the best phenotype.
An Inducible Toxin
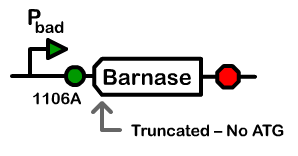
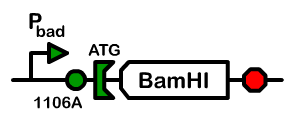
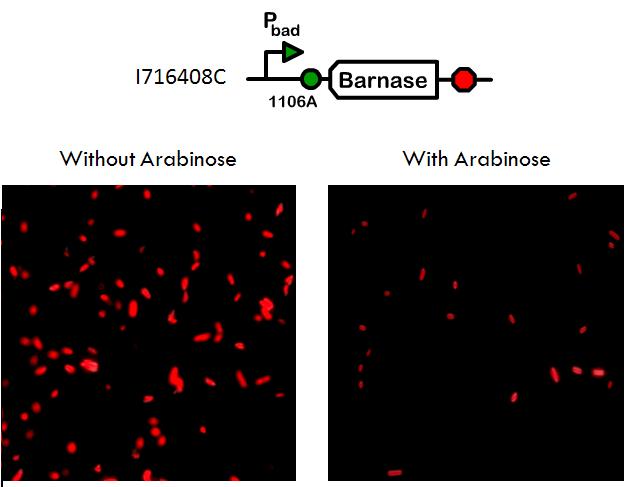
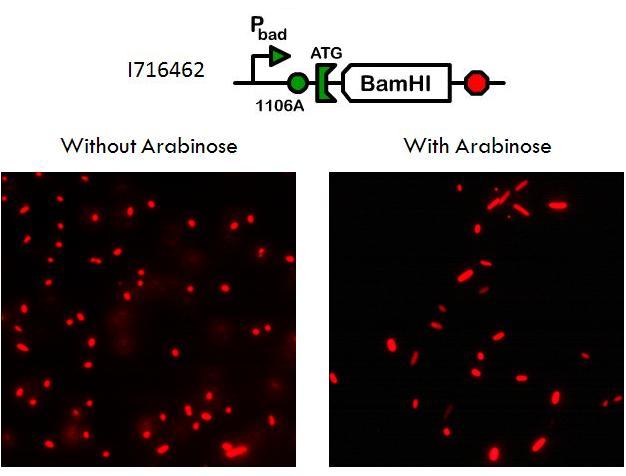
Using a pBad promoter, we constructed several variations of inducible toxins, including the dnase ceaB from the coilicin activity protein, endonucleases BamHI and BglII, and rnase barnase. Additionally, ribosome binding site libraries were used in order to increase the likelihood of finding a construct that would exhibit no growth after being induced with arabinose, but normal growth when uninduced.
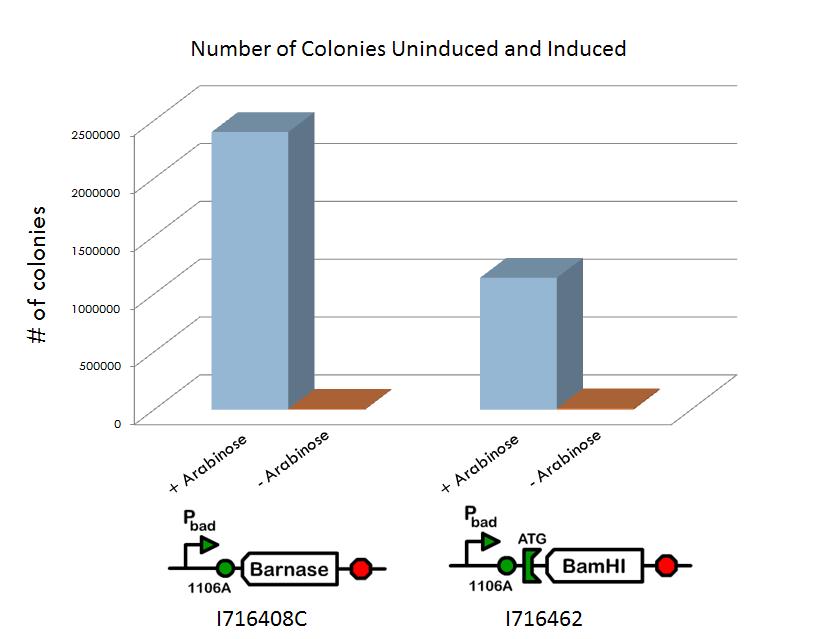
We screened libraries of potential hits with a tecan growth assay, and in the end, the constructs that showed the desirable phenotype are the constructs shown below (I716408C and I716462). When uninduced, the cells showed growth comparable to regular DH10B cells, but when induced, the growth plateaus as the cells lose their ability to replicate due to RNA and DNA destruction.
Characterization of Growth Ability
Additional characterization of the systems include a titer experiment, where the cells were grown up in a cultures both with and without arabinose. The cultures were then diluted and plated such that the number of colonies could be counted. It was shown that the cultures induced with arabinose had significantly fewer colonies than those not induced with arabinose, after taking into account the various dilutions.
Phenotype of dead cells
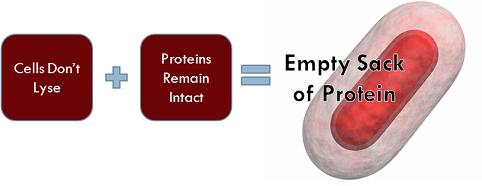
Importantly, although the cells triggered with arabinose have lost their ability to reproduce, they do not lyse, because if the cells did not remain intact, then they could not serve the purpose of transporting hemoglobin. Equally significant is the fact that the the proteins within the cell are not degraded and still maintain their function.
This phenotype was confirmed by transforming an RFP plasmid into the cell and then taking microscopy pictures of the cells grown up in cultures with and without arabinose. With microscopy, it is clear that the cells are still capable of expressing the RFP proteins even when their growth has been arrested.