Harvard
From 2007.igem.org
(→Quorum Sensing) |
(→Quorum Sensing) |
||
| Line 85: | Line 85: | ||
</gallery> | </gallery> | ||
'''Conclusions and future plans''' | '''Conclusions and future plans''' | ||
| - | *Continue characterization single cell constructs (approach (1) ). | + | *Continue characterization single cell constructs (approach (1) under Project Overview). |
*Characterize cell and bead density require to reach quorum levels. We have been attempting to do this through Nickel-coated 96 well plates. | *Characterize cell and bead density require to reach quorum levels. We have been attempting to do this through Nickel-coated 96 well plates. | ||
* | * | ||
Revision as of 20:49, 23 October 2007
| Harvard University 2007 iGEM Team | ||
Students
Education Advisor
|
Teaching Fellows
Faculty Advisors
| |
The Harvard University 2007 iGEM Team
This year Harvard's team consisted of eight undergraduate students, with backgrounds in molecular and cellular biology, biochemistry, engineering, and computer science. With the help of six faculty advisers and four teaching fellows, plus one education advisor, they devised and executed a single project with three subsections.
Cling-E. Coli
As of the end of the summer, we successfuly demonstrated enriched targeting with known tagged sender and receiver cells. Separately, we characterized the quorum system with multiple types of promoters and reporters. We are on our way toward a combined targeting-quorum system in the near future. In addition, targeting with random libraries to various substrates should also be achieved.
Eventually, our project may have applicability in medical imaging and localized drug synthesis and release. Theoretically, if our project is successful, we demonstrate that bacteria (or other microorganisms) can be targeted specifically to a certain tumor cell. At a certain density, quorum sensing may allow for the bacteria to emit a signal that can be detected by medical imaging devices. If the reporter gene encoded for drugs or vitamins, synthesis can also be evoked through enriched targeting. Furthermore, binding through a modified Fec system may allow for binding alone to transduce a signal and elicit a response in the cells.
Bacterial Targeting
Project Overview
- The motivation for this part of the project was to engineer bacteria to adhere to targets with a high degree of specificity.
- Initial targeting was done by displaying histidine and strep2 tags on the E. coli surface via fusion with LppOmpA and AIDA-1 vehicles, and screens were performed with binding to their known nickel and streptavidin targets, respectively.
- After characterization and high enrichment with these known substrates, random libraries were inserted into LppOmpA and AIDA-1 constructs for screening peptides with affinity for novel targets. As we proceed with this experiment, we hope to characterize sequences that lend specificity to calmodulin and EGF.
- We also tried to utilize the same enrichment strategy with cells that were cotransformed with both AIDA-1-fused tags and quorum-sensing genes (see below).
Methods
Results
Conclusions and future plans
References
Quorum Sensing
Project Overview
- The motivation for this part of the project was to effect downstream activity after E. coli bind to a particular substrate, using the luxI/luxR quorum-sensing system from Vibrio fischeri, which would turn on after the bacteria localize to the target.
- Lux quorum-sensing works like a system of sender and receiver. LuxI codes for a protein that catalyzes the synthesis of 3-oxo-hexanyl homoserine lactone, or OHHL, which can diffuse freely through the surface of E. coli into the environment and into other E. coli. LuxR encodes for a protein which does not diffuse across the membrane and, when bound to OHHL, upregulates the luxpR promoter. So luxI-producing bacteria (senders) produce OHHL that diffuses into luxR-producing bacteria (receivers) where OHHL and luxR turn on luxpR promoter. This only occurs at a certain concentration of OHHL, thus a certain concentration of cells (quorum) is required.
- Initial characterizations of the luxI/luxR system and quorum activity were made using GFP and RFP reporters. Two approaches were taken to quorum sensing. (1) A luxI/luxR production system in one cell would be simpler, but it's possible that the cells might self-induce. (2) luxI and luxR production in separate cells would ensure no self-induction, but it requires monitoring two populations of cells.
Methods
- To characterize quorum activity, constitutive RFP senders were mixed with constitutive receivers. Fluorescence and OD (cell density) readings were taken every 15 minutes. [http://openwetware.org/wiki/IGEM:Harvard/2007/Laboratory_Notebooks/Quorum_Sensing/Plate_Reader_Protocol The protocol] was adjusted as necessary according to what the exact desired experiment was.
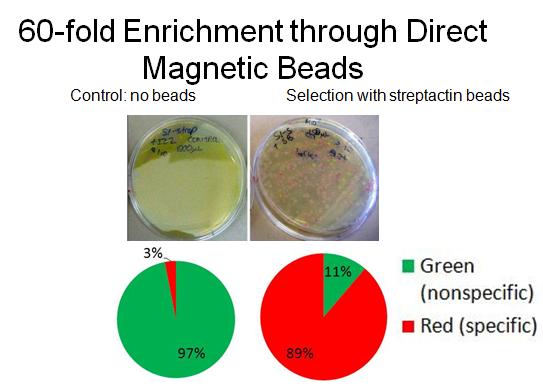
- To characterize binding activity, RFP-senders with AIDA tags were mixed with constitutive GFP non-tagged cells. As a control, a portion of the mixed cells was plated pre-selection. Direct magnetic beads (TALON nickel, Qiagen streptactin) were used to enrich for the tagged-senders. Several washes were done in low concentration imidazole. After incubations with beads and washes, the mixture was plated and grown overnight, and cell counts were done to determine extent of enrichment. (A more detailed protocol [http://openwetware.org/wiki/Shlo/directmagneticprotocol available]).
- More detailed methods are available on our [http://openwetware.org/wiki/IGEM:Harvard/2007/Laboratory_Notebooks/Quorum_Sensing wiki].
Results
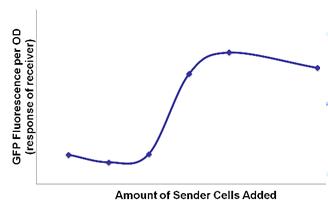
- We were able to demonstrate that the sender-receiver system in one cell was not self-inducing but acted with quorum activity.
- Quorum activity was also detected with the separate sender and receiver cells.
- In collaboration with the [http://openwetware.org/wiki/IGEM:Harvard/2007/Bacterial_Targeting bacterial targeting] subteam, we have evidence that red sender cells tagged with strep2 on the surface (cotransformed with constitutive RFP/luxI plasmid and AIDA-strep2 plasmid) are enriched by selection with streptavidin-coated beads, while green non-tagged cells are not.
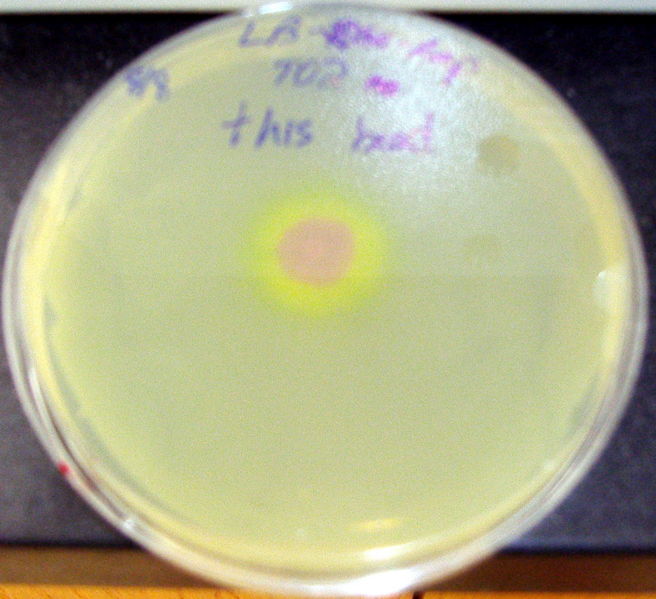
- Quorum activity was demonstrated as well through a plate-drop experiment.
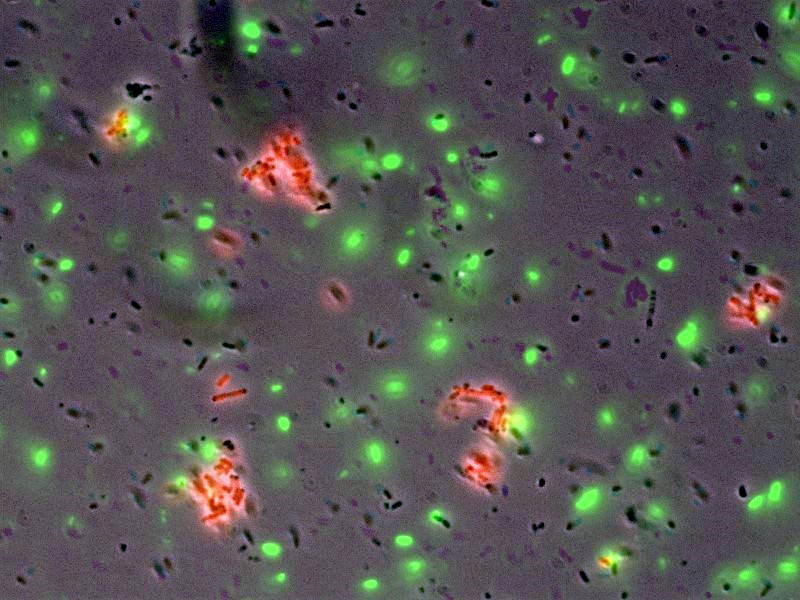
- [http://openwetware.org/wiki/Shlo/flmicroprotstickiness Microscopy] was also used to detect and image tagged bacteria accumulating around beads, while non-tagged bacteria did not aggregate.
Conclusions and future plans
- Continue characterization single cell constructs (approach (1) under Project Overview).
- Characterize cell and bead density require to reach quorum levels. We have been attempting to do this through Nickel-coated 96 well plates.
References
- Voigt CA. Genetic parts to program bacteria. Curr Opin Biotechnol 2006 Oct; 17(5) 548-57. doi:10.1016/j.copbio.2006.09.001 pmid:16978856.
- Anderson JC, Clarke EJ, Arkin AP, and Voigt CA. Environmentally controlled invasion of cancer cells by engineered bacteria. J Mol Biol 2006 Jan 27; 355(4) 619-27. doi:10.1016/j.jmb.2005.10.076 pmid:16330045
Fec Signal Transduction
Project Overview
- The motivation for this part of the project was also to effect downstream activity after E. coli binds to a target, but through signal transduction of the Fec system.
- Fec System Overview:
- The Fec system consists of outer membrane protein FecA. Its wild-type ligand is ferric citrate.
- When binding occurs in iron-limiting conditions, a signal is sent to the inner membrane protein FecR.
- FecR activates sigma factor FecI.
- FecI is then used to induce gene expression.
- The Fec system of E. coli is unique in its ability to bind to iron and produce a signal to control gene expression. We proposed two options to modify the binding of FecA and hopefully maintain signalling:
- 1) A computational approach which would produce nucleotide sequences for binding our new target. A directed mutagenesis of FecA would follow.
- 2) Inserting a binding sequence into loop 7 of FecA such that it would bind to our new target at least, and produce a signal at most. The binding sequences chosen are 6xHis and Strep, mainly as simple proofs-of-concept.
Methods
- The main constructs used were shipped from the Braun group in Germany. These include AA93 E.Coli strain which does not express the Fec system, a GFP plasmid on a Fec promotor, and pLCIRA, a plasmid that for Fec expression. We cut out the loop 7 section of the FecA gene and re-inserted a modified version with 6xHis or Strep inserted.
- Because pLCIRA is not well characterized, we also attempted to use a T7 pCola duet vector which has 2 multiple cloning sites. We inserted FecIR (pcr'ed from the pLCIRA) into the first cloning site and FecA (also pcr'ed from pLCIRA) into the second. However, AA93 is not a T7 cell, so we used lysogenization to make AA93 DE3 cells.
For more details, see the Harvard iGEM page...
Results
Conclusions and future plans
References
Click the picture, or click [http://openwetware.org/wiki/IGEM:Harvard here] for the more detailed Harvard iGEM 2007 wiki.