Paris/DesignProcess
From 2007.igem.org
Eismoustique (Talk | contribs) (→Overview of the project) |
Eismoustique (Talk | contribs) (→Optimization through feedback) |
||
| Line 102: | Line 102: | ||
====Optimization through feedback==== | ====Optimization through feedback==== | ||
| - | It is clear that the average recombination rate must be between 0 and 50% per generation. Nevertheless, we can theoretically maximize the growth rate by adjusting the recombination rate to | + | It is clear that the average recombination rate must be between 0 and 50% per generation. Nevertheless, we can theoretically maximize the growth rate by adjusting the recombination rate to DAP level as shown by our models. In this way, the germ line would increase its differentiation rate upon DAP starvation, and reduce it when there is enough DAP. In order to achieve this, Cre expression must be adjusted to DAP concentration. This would be easily done if there where a promoter sensitive to DAP concentration, which seems to be the case of the dapA promoter (dapAp). We therefore cloned and characterized this promoter. |
| Line 111: | Line 111: | ||
| - | |||
| - | |||
| - | Through classical molecular biology techniques, we aimed | + | Through classical molecular biology techniques, we aimed to construct the following genomically inserted cassette. |
| + | To learn more about the construction process, [[Paris/ConstructionProcess|click here]] or follow the link at the end of the page. | ||
| - | |||
| - | + | [[Image:Paris_FinalConstruct.JPG|500px]] | |
This full construct has yet to be assembled in the genome. | This full construct has yet to be assembled in the genome. | ||
Revision as of 21:03, 25 October 2007
Contents |
SMB Design Process
A first question that needs to be addressed in the design process of our system is that of the nature of the dependency relationships between its cellular components. Namely, what cross-dependency relationship between the somatic & germ line cells can best ensure “ecological” equilibrium & “evolutionary” stability between the two cell types?
The soma needs the germ line
What dependency relationship?
A radical mean of ensuring somatic cell dependence on germ line is to make somatic cells sterile. That is, if somatic cells are unable to replicate, then they exclusively depend on G=>S differentiation for their existence. Furthermore, in this way it is impossible for S cells to overtake the population.
How can sterility be achieved?
A great number of essential genes have been identified since the early days of E.coli genetics. An essential gene is one for which a mutant strain cannot grow at 37° on rich medium (LB). These genes include the essential division genes. Mutants of this genes cannot divide. Suppressing the expression of such a gene would thus make the cell sterile.
Differentiation mechanism
As discussed above, differentiation can be epigenetic (changes in genes expression pattern) or genetic (change in DNA sequence). In order to suppress the expression of an essential gene, we could either place it under the control of an inducible promoter that we would repress (epigenetic differentiation), or remove the gene itself through recombination (genetic differentiation). A genetic mechanism of differentiation is probably more robust than an epigenetic one in a way that the system artificially implemented in the cell should escape with more difficulties.
Therefore, we decided that the Germen=>Soma differentiation mechanism should be genetic (as opposed to epigenetic). Sterility in S cells will be achieved by deleting an essential gene from the genome during the differentiation process. This will be done through site-specific recombination.
Constraints on essential gene choice
Remains the question of the choice of the essential gene to be used. We have decided to base our choice on two criteria:
- Longevity of the S cells: S cells even if unable to divide should be able to live as long as possible. (an explanation for this is given below)
- Gene isolation: In our final construct, cassettes will be inserted before and after the essential gene. We want our gene to be isolated, so as not to disturb the expression of other nearby genes. It should not for instance be embedded in an operon. See here for more details about the construction method.
We have finally chosen ftsK as the gene to be used. It is an isolated gene. Also, ts (thermo sensitive) strains for this gene give filamentous cells. The cells continue to grow without dividing until they die. This can go on for more than 10 generations, and the cells can reach impressive sizes.
The germ line needs the soma
What dependency relationship?
In our synthetic organism, the germ line is auxtroph for a given nutrient that is provided by the soma. In this way, a part of the germ line needs to differentiate, in order to feed the undifferentiated fraction.
Why an auxotrophy dependence?
Previous works have shown that two types of cell, each auxotroph for a different metabolite, can rescue each other when grown in the same minimum media. This means that some auxotrophy should be rescued in a coculture with prototroph cells. Plus, it is very easy to do!
What auxotrophy?
In order to choose our auxotrophy, we need to take several constraints into account:
- No simple bypass or reversion: The auxotrophy phenotype of the germ line must be stable. There must be no simple metabolic bypass or reversion possibilities.
- Overproduction and excretion: Prototroph cells must be able to overproduce and excrete the metabolite, or at least to release it when dying.
- Absorption: auxotroph cells should be able to live in very low concentration of this metabolite. Indeed we should expect only little metabolite to be excreted.
- Survival: auxotroph cells should be able to live as long as possible when deprived from the metabolite.
- No growth on LB: We would like our synthetic organism to be able to grow on rich medium. This would facilitate our lab work, and more generally allow our synthetic organism to grow in a variety of conditions.
- Feedback: For purposes described below, we would like to have a sensor device sensitive to our metabolite concentration.
We finally choose diaminopimelate (DAP) as our metabolite. The germ line will be deleted for the dapA gene, an essential gene in the peptidoglycan and lysine biosynthesis pathways. For details about our choice see here.
In order to confirm that our choice fits well the above mentioned criteria, we conducted several preliminary experiments. (See the results page)
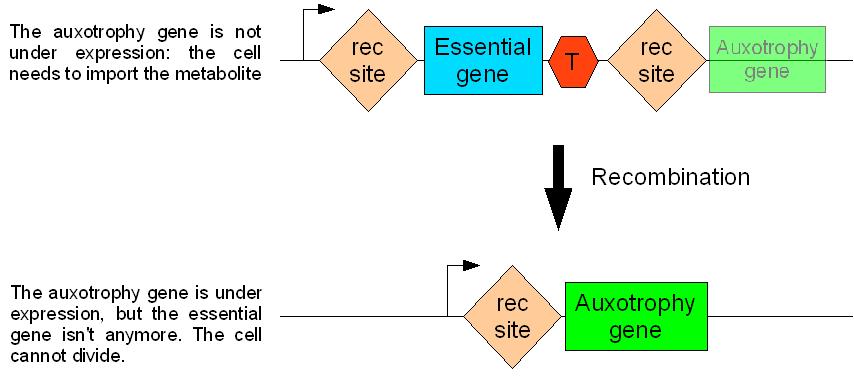
How will auxotroph germ line cells differentiate into prototroph somatic cells?
We found an elegant way to solve this problem. This is best described by the following schema.
rec is a site specific recombination sequence
T is a strong terminator
System scale specifications
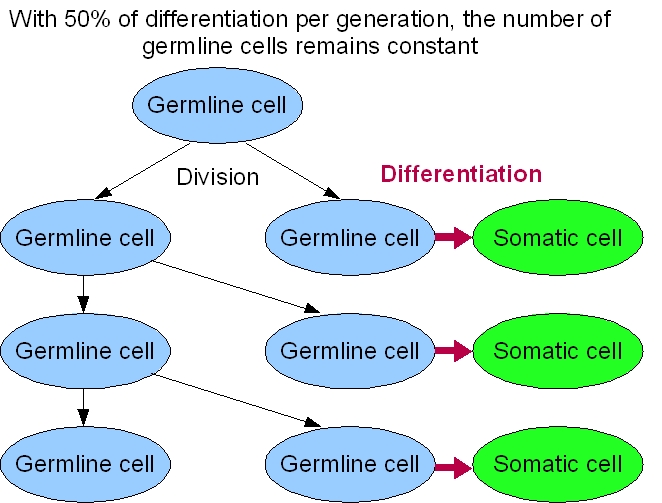
What differentiation rate?
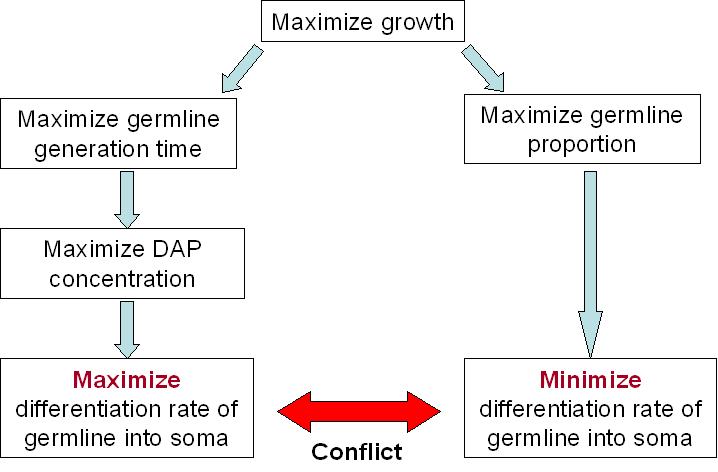
Our system's success relies a lot on the differentiation rate of the germ line into soma. An intuitive reasoning shows that we cannot have more than 50% of differentiation per generation. Otherwise, the germ line will only decrease. But this is not the only constraint to take into account.For our germ line to grow fast, we need to maximize the DAP production. This means that we need to maximize the excretion of DAP by the soma and to maximize the proportion of the soma itself. The number of somatic cells is given by the differentiation rate of germ line cells and the life time of somatic cells. Thus we need to maximize the life time of somatic cells and the differentiation rate of the germ line. On the other hand, we want our synthetic organism to grow as fast as possible. And it will grow faster if the proportion of germ line cells is bigger, since somatic cells do not divide. This means that we have to minimize the differentiation rate.
We clearly see the conflict here, which will ultimately lead to a tradeoff between the germ line proportion and its generation time. As explained above, the differentiation rate cannot be over 50% per generation if we want our synthetic organism to grow. What this tradeoff tells us is that there is an optimum differentiation rate somewhere between 0 and 50% recombinant per generation. This is best illustrated by the modeling of our system.
What recombination system?
According to the previous point, we need a recombination system whose rate can be controled. Ideally we wish to obtain recombination at any chosen frequency. If this is impossible, we need at least the recombination rate to be below 50% per generation, but high enough for the germ line to be sufficiently fed. Another constraint on the recombination system, is that we want differentiation to be unidirectional. We thus chose the Cre/Lox recombination system, for the following reasons:
- it is readily available, largely used and well described
- lox66 and lox71 recombination sites have been described to produce unidirectional recombination ( Cre recombinase-mediated inversion using lox66 and lox71: method to introduce conditional point mutations into the CREB-binding protein, Zuwen Zhang and Beat Lutz, Nucleic Acids Research)
We want to control the recombination frequency by adjusting the expression level of the Cre recombinase. To do this, we cloned the Cre recombinase under the control of the pBad promoter (inducible through arabinose). We also decided to clone our Cre production device (araC-pBad>>rbs-Cre) on a low copy number plasmid, to broaden the reachable expression rates.
Recombinaison frequency measurment experiments
Optimization through feedback
It is clear that the average recombination rate must be between 0 and 50% per generation. Nevertheless, we can theoretically maximize the growth rate by adjusting the recombination rate to DAP level as shown by our models. In this way, the germ line would increase its differentiation rate upon DAP starvation, and reduce it when there is enough DAP. In order to achieve this, Cre expression must be adjusted to DAP concentration. This would be easily done if there where a promoter sensitive to DAP concentration, which seems to be the case of the dapA promoter (dapAp). We therefore cloned and characterized this promoter.
Overview of the project
(Flash animation J-3)
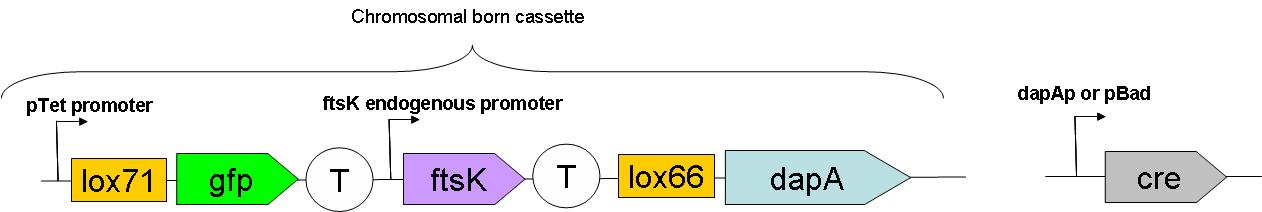
Through classical molecular biology techniques, we aimed to construct the following genomically inserted cassette.
To learn more about the construction process, click here or follow the link at the end of the page.
This full construct has yet to be assembled in the genome.
As can be seen on this schematic representation of the SMB genomic backbone cassette, at basal genomic state (in Germ line cells):
- pTet promoter drives the expression of gfp.
- The expression of ftsK is controlled by its natural promoter.
- ftsK is isolated from pTet promoter by the intercalation of Terminator (B0015 terminator).
- dapA gene is in a dormant state since it lacks a promoter to drive its transcription.
Upon G to S differentiation, the following genomic reassembly should take place:
constructed. These are termed the “upstream construct” & the “downstream construct”:
Cre mediated lox recombination should lead to:
- excision of ftsK gene from the genome on a circular unreplicative DNA molecule. We ignore how much time such a DNA is stable in a cell.
- placing dapA gene under the control of pTet promoter. This should lead to dapA expression, & hopefully to DAP synthesis.
E. Colight: towards a new slim diet
Project
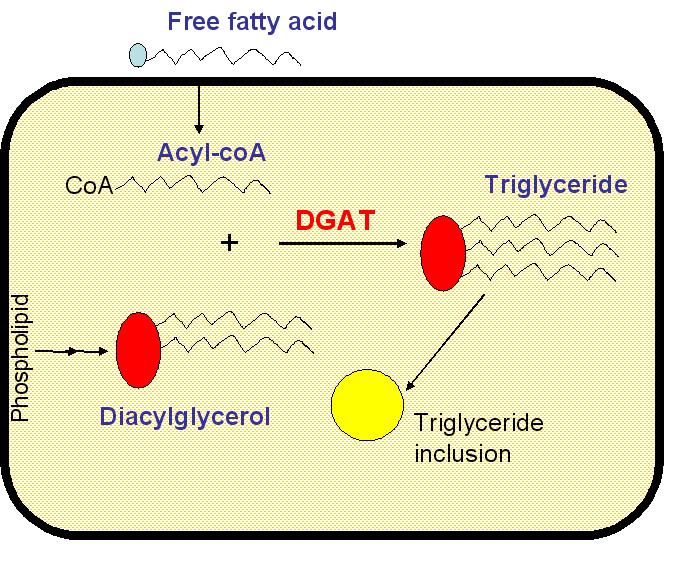
Triglycerides are composed of a molecule of glycerol esterified by three fatty acids. When ingested, triglycerides are hydrolysed by lipases, in the stomach and the duodenum, into glycerol and free fatty acids. Enterocytes are can only absorb free fatty acids and glycerol. These are subsequently recombined in the cytoplasm into triglycerides. Triglycerides are then freed in the lymphatic system and then in the blood within fatty vesicles called chylomicrons.
When lipid uptake versus energy consumption and loss is unbalanced, we accumulate fat. Knowing that gut is full with bacteria forming the gut microflora (we have 1013 cells in our body and 1014 bacteria with the majority in the gut!), we envision engineering bacteria capable of absorbing fatty acids and storing them in the form of triglycerides intracellular inclusion. These triglycerides would not be absorbed by enterocytes! Lipid input would decrease! Eat fat, don't get fat!
In fact, a drug, orlistat, already exists and shows decreasing lipid input works in obesity. It comes from a bacterial lipase inhibitor (from Streptomyces toxytricini). It inhibits pancreatic lipase and is used to cure obese people and type 2 diabetes with hypocaloric cure. At the standard prescription dose of 120 mg three times daily before meals, orlistat prevents approximately 30% of dietary fat from being absorbed (Thomson PDR, 2006).
E.coli is the most used bacterium in synthetic biology and... belongs to the gut microflora! We wish to genetically engineer E. coli into Ecolight to store triglycerides into inclusions! Knowing that 40% of E.coli is renewed every day, these triglyceride-filled bacteria will leave the gut with faeces!
The result of this work should, in a second phase of our work, be combined with the “security device” derived from the SMB described above. This is further discussed in PERSPECTIVES.
Technical details
As a perspective for our SMB, we want to show that it can be a tool for metabolic engineering (see perspectives). We started to develop the idea of making E. Coli store fatty acids in the form of triglycerides. Such an engineered bacteria could be ingested to absorb fatty acids thus limiting the amount of fatty acids absorbed by the user! Eat fat don't get fat !
- Why should it work?
Triglycerides are not a natural product of E. coli metabolism as it lacks diacylglycerol acyl-transferase (DGAT). DGAT catalyses the reaction of glycerol esterifaication by fatty acids. However, all the compounds necessary for the triglyceride synthesis are present in the cytoplasm: diacylglycerol is an intermediate of phospholipid catabolism (source ecocyc.org) and free fatty acids are imported from extracellular medium. Wild-type E. coli strains can indeed grow on free fatty acid medium (oleate for example). E. Coli has a Long Chain Fatty Acid (LCFA) transporter, FadL. Another protein: FadR, a long chain acyl-CoA-responsive transcription factor, controls the expression of nine genes primarily involved in fatty acid degradation and biosynthesis (FadL is thus induced by FadR) and β-oxidation enzymes.
A qualitative model of E.coli metabolism (Palsson) has shown ????? Qu'est-ce qu'il a montré exactement????.
- Which enzyme?
We decided to use DGAT enzyme imported from a bacterium closely related to E. coli for metabolic compatibility (je comprend pas?????????). Acinetobacter calcoaceticus ADP1, a Gram negative bacillus was a good candidate. Its DGAT enzyme is also an acyl-CoA fatty alcohol acyltransferase (wax ester synthase) and catalyzes the final condensation of acyl-CoA and fatty alcohol. But knowing that E.coli does not produce fatty alcohol, this reaction is probably not avaible. (for more information see BBa_I718002).