BerkiGEM2007Present1
From 2007.igem.org
(→Heme) |
|||
| Line 1: | Line 1: | ||
<html> | <html> | ||
<b><a href="https://2007.igem.org/Berkeley_UC"><< Back to UC Berkeley iGEM 2007</a> <font face="Arial">•</font> <a href="https://2007.igem.org/BerkiGEM2007Present4">Next: The Chassis >></a></b><br /></html> | <b><a href="https://2007.igem.org/Berkeley_UC"><< Back to UC Berkeley iGEM 2007</a> <font face="Arial">•</font> <a href="https://2007.igem.org/BerkiGEM2007Present4">Next: The Chassis >></a></b><br /></html> | ||
| + | |||
| + | ==Hemoglobin== | ||
| + | Hemoglobin: | ||
| + | • Basics of wild type hemoglobin. (basic descriptions) | ||
| + | (Image of hemoglobin cassette) | ||
| + | The primary component needed for efficient oxygen transport in our system is human hemoglobin A. (HbA) HbA is a tetramer that consists of two different subunits, α2β2. | ||
| + | |||
| + | |||
| + | • Problems of using free hemoglobin as a blood substitute. | ||
| + | There are several problems which we have addressed with regards to hemoglobin and oxygen transport. | ||
| + | Problems: | ||
| + | Insufficient P50 of wild type human hemoglobin. | ||
| + | The first of which is the insufficiently low P50, the partial pressure of oxygen needed for 50% saturation, of wild type human hemoglobin A. The P50 for wild type human hemoglobin is ~3.8 at physiological conditions. | ||
| + | In human erythrocytes, this problem is fixed primarily by the presence of an allosteric modifier 2,3-diphosphoglycerate (2,3-DPG) which forces the hemoglobin conformation into the lower affinity deoxy state, or the T-state. By pushing the hemoglobin into the T-state, 2,3-DPG is effectively pushing any bound oxygen out of the bound state and decreasing the oxygen binding affinity of the hemoglobin. This decreased oxygen binding affinity causes the P50 to be much higher than if the allosteric modifier were not present. An increase of 0.4mM in DPG concentration decreases oxygen affinity by about 1.0 torr and the normal concentration of DPG in erythrocytes is ~5mM. | ||
| + | (Image of P50 curves identified for each mutant and wild type) | ||
| + | Our solution | ||
| + | In our system we have chosen to use well studied mutants of human hemoglobin which have been engineered to be permanently in the deoxy T-state. The mutations are named Presbyterian (beta-Asn108Lys) and providence (beta-Lys82Asp). | ||
| + | The P50 is also sensitive to various ions such as Cl-, although to a much smaller amount than 2,3-DPG. The portion of hemoglobin that binds these types of modifiers is at the N-terminal end. When hemoglobin is expressed in E. coli, there is an extra methionine residue which is normally cleaved off in eukaryotic cells. | ||
| + | |||
| + | Precipitation of alpha-HbA and decreased yield of functional hemoglobin. | ||
| + | The alpha subunit is more prone to precipitation because an alpha-alpha dimer is insoluble under normal conditions. This can cause an excess of beta subunits and decreased functional hemoglobin output. | ||
| + | |||
| + | The solution in erythrocytes is to use a chaperone named alpha hemoglobin stabilizing protein (AHSP), which binds to single alpha subunits and facilitates the transfer of a beta subunit to the alpha. An alpha-beta dimer remains soluble. | ||
| + | Our use of the bacterial chassis as a container that mimics an erythrocyte allows us to provide the same solution as in an erythrocyte. We include in our system the gene that encodes for human AHSP. In addition, we have also explored a fusion of di-alpha subunits with a glycine linker. This has been proven to give a higher soluble output by somehow stabilizing the alpha subunits and preventing precipitation. | ||
| + | |||
| + | Autoxidation of the heme center causes free radical formation | ||
| + | During the process of binding and unbinding oxygen, the four heme groups of the hemoglobin may spontaneously undergo autoxidation, ultimately causing the formation of damaging free radicals. This causes two problems. The first problem is that the free radicals can cause damage to cellular proteins, affecting their function. Another problem is that when autoxidation occurs, an electron is transferred from the Fe2+ iron center to the oxygen, creating a superoxide and leaving the heme with a Fe3+ metal center. The resulting hemoglobin is known as methemoglobin and it is unable to carry oxygen. The autoxidation occurs to a significant amount on the order of hours. | ||
| + | Erythrocytes remedy the problem of free radical accumulation and damage by containing the antioxidant enzymes catalase and superoxide dismutase. These enzymes catalyze the breakdown of superoxide into oxygen and H2O. | ||
| + | Human erythrocytes have addressed the autoxidation problem by using the NADH dependent enzyme, cytochrome b5 reductase. The basic mechanism is shown here. | ||
| + | |||
| + | |||
| + | |||
== Heme == | == Heme == | ||
| Line 45: | Line 77: | ||
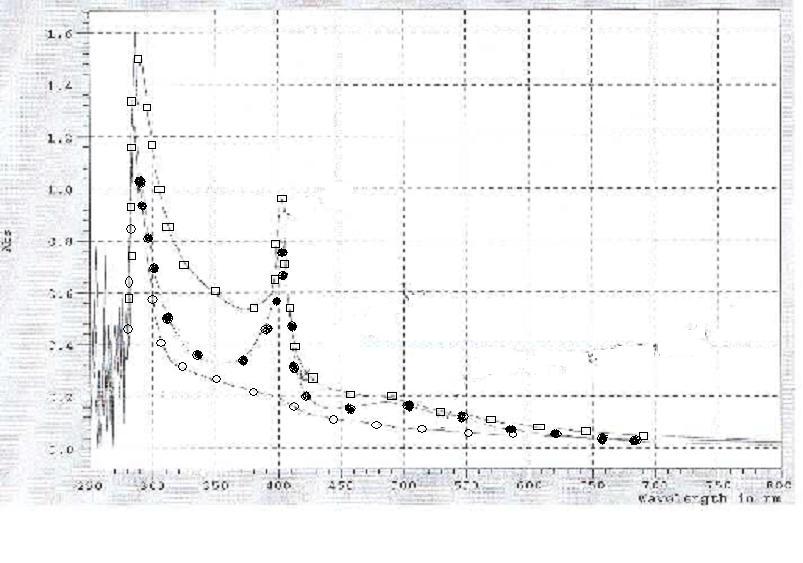
With our construct complete, we employed UV-Vis to confirm that the red product we are seeing is, in fact, heme. The maximum absorbance for heme occurs at 412 nm. As the graph shows, the hemA from both R.capsulatus and CFT073 combined with the hemB have significant peaks at wavelengths corresponding to the presence of heme, 412nm. | With our construct complete, we employed UV-Vis to confirm that the red product we are seeing is, in fact, heme. The maximum absorbance for heme occurs at 412 nm. As the graph shows, the hemA from both R.capsulatus and CFT073 combined with the hemB have significant peaks at wavelengths corresponding to the presence of heme, 412nm. | ||
| - | |||
| - | |||
| - | |||
== Alternatives To Hemoglobin == | == Alternatives To Hemoglobin == | ||
Revision as of 04:02, 26 October 2007
<< Back to UC Berkeley iGEM 2007 • Next: The Chassis >>
Hemoglobin
Hemoglobin: • Basics of wild type hemoglobin. (basic descriptions) (Image of hemoglobin cassette) The primary component needed for efficient oxygen transport in our system is human hemoglobin A. (HbA) HbA is a tetramer that consists of two different subunits, α2β2.
• Problems of using free hemoglobin as a blood substitute.
There are several problems which we have addressed with regards to hemoglobin and oxygen transport.
Problems:
Insufficient P50 of wild type human hemoglobin.
The first of which is the insufficiently low P50, the partial pressure of oxygen needed for 50% saturation, of wild type human hemoglobin A. The P50 for wild type human hemoglobin is ~3.8 at physiological conditions.
In human erythrocytes, this problem is fixed primarily by the presence of an allosteric modifier 2,3-diphosphoglycerate (2,3-DPG) which forces the hemoglobin conformation into the lower affinity deoxy state, or the T-state. By pushing the hemoglobin into the T-state, 2,3-DPG is effectively pushing any bound oxygen out of the bound state and decreasing the oxygen binding affinity of the hemoglobin. This decreased oxygen binding affinity causes the P50 to be much higher than if the allosteric modifier were not present. An increase of 0.4mM in DPG concentration decreases oxygen affinity by about 1.0 torr and the normal concentration of DPG in erythrocytes is ~5mM.
(Image of P50 curves identified for each mutant and wild type)
Our solution
In our system we have chosen to use well studied mutants of human hemoglobin which have been engineered to be permanently in the deoxy T-state. The mutations are named Presbyterian (beta-Asn108Lys) and providence (beta-Lys82Asp).
The P50 is also sensitive to various ions such as Cl-, although to a much smaller amount than 2,3-DPG. The portion of hemoglobin that binds these types of modifiers is at the N-terminal end. When hemoglobin is expressed in E. coli, there is an extra methionine residue which is normally cleaved off in eukaryotic cells.
Precipitation of alpha-HbA and decreased yield of functional hemoglobin. The alpha subunit is more prone to precipitation because an alpha-alpha dimer is insoluble under normal conditions. This can cause an excess of beta subunits and decreased functional hemoglobin output.
The solution in erythrocytes is to use a chaperone named alpha hemoglobin stabilizing protein (AHSP), which binds to single alpha subunits and facilitates the transfer of a beta subunit to the alpha. An alpha-beta dimer remains soluble. Our use of the bacterial chassis as a container that mimics an erythrocyte allows us to provide the same solution as in an erythrocyte. We include in our system the gene that encodes for human AHSP. In addition, we have also explored a fusion of di-alpha subunits with a glycine linker. This has been proven to give a higher soluble output by somehow stabilizing the alpha subunits and preventing precipitation.
Autoxidation of the heme center causes free radical formation During the process of binding and unbinding oxygen, the four heme groups of the hemoglobin may spontaneously undergo autoxidation, ultimately causing the formation of damaging free radicals. This causes two problems. The first problem is that the free radicals can cause damage to cellular proteins, affecting their function. Another problem is that when autoxidation occurs, an electron is transferred from the Fe2+ iron center to the oxygen, creating a superoxide and leaving the heme with a Fe3+ metal center. The resulting hemoglobin is known as methemoglobin and it is unable to carry oxygen. The autoxidation occurs to a significant amount on the order of hours. Erythrocytes remedy the problem of free radical accumulation and damage by containing the antioxidant enzymes catalase and superoxide dismutase. These enzymes catalyze the breakdown of superoxide into oxygen and H2O. Human erythrocytes have addressed the autoxidation problem by using the NADH dependent enzyme, cytochrome b5 reductase. The basic mechanism is shown here.
Heme
Introduction
Heme is a prosthetic group to hemoglobin. Generally, heme consists of an iron atom surrounded by a porphyrin ring. Each hemoglobin molecule is capable of binding up to four heme groups. One of the most important functions of heme is to assist in the transportation of diatomic gases. In red blood cells, heme and hemoglobin are the components that allow the binding of oxygen. As a result of this interaction, red blood cells continuously deliver oxygen to the entire body.
The Pathway
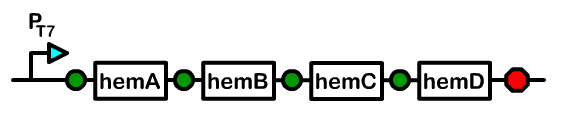
The biosynthetic pathway for heme demonstrates that there are eight enzymes, or heme genes, involved in the production of heme. Although bacteria already have these genes present in their genome, the amount of intermediates produced are insufficient for our purposes. We worked solely with hemA (Delta-aminolevulinate synthase), hemB (Delta-aminolevulinic acid dehydratase), hemC (porphobilinogen deaminase), and hemD (uroporphyrinogen III synthase) because over-accumulation of heme in our cells would result in toxicity. After successful subcloning experiments, the bacterial cell pellets would become reddish-brown due to the accumulation of porphyrins.
Design and Construction
First, we cloned hemA from R.capsulatus and hemA from CFT073, not knowing which hemA from which organism would give us the highest yield of heme. We also cloned hemB, hemC, and hemD from MG1655. Afterwards, we attached single ribosomal binding sites as well as library ribosomal binding sites. The advantage of working with library ribosomal binding sites is after transformation, one can grow single clones in a 96-well block and selectively choose the clone producing the greatest amount of heme, which correlates with the clone yielding the most red of pellets.
As depicted in the image to the left, because of the phenotype of porphyrins and heme, it was easy to select single colonies that combined with the strongest ribosomal binding sites of the library. These stronger clones would yield the greatest amount of heme and have the deepest red/brown color of all the clones.
After the ribosomal binding sites were attached to the heme genes, the genes were subcloned onto the same plasmid in sequential order. We placed the T7 promoter we produced at the beginning of the composite part as well as a terminator at the end.
With our construct complete, we employed UV-Vis to confirm that the red product we are seeing is, in fact, heme. The maximum absorbance for heme occurs at 412 nm. As the graph shows, the hemA from both R.capsulatus and CFT073 combined with the hemB have significant peaks at wavelengths corresponding to the presence of heme, 412nm.
Alternatives To Hemoglobin
Over the summer I investigated and created two alternatives to the hemoglobin part in our device. I worked with two genes we believed might allow our blood to carry more oxygen than hemoglobin.
The first gene I will discuss is H-NOX (Heme-Nitric oxide and Oxygen bonding). H-NOX is a heme-based sensor that is found in bacteria. H-NOX is able to bond to Oxygen using a distal pocket tyrosine. For this gene I added the T7 promoter we created for this project, an RBS site, and lastly a Bca1092 terminator. As I said previously this part would replace the hemoglobin part in our final device. When I assayed this part the results were inconclusive. The part was assembled correctly but the assay didn't show strong signs of expression.
The second gene I explored was Sperm Whale Myoglobin. Myoglobin is a monomeric protein that behaves as an intracellular oxygen storage site. Sperm whale myoglobin in particular is easily found in large amounts in the whale's muscle tissue. The construction of this part was very similar to that of the H-NOX composite part. Used the same promoter, terminator, and RBS. The assay for Myoglobin showed a bit more promise but couldn't conclusively show that Myoglobin was binding to oxygen.