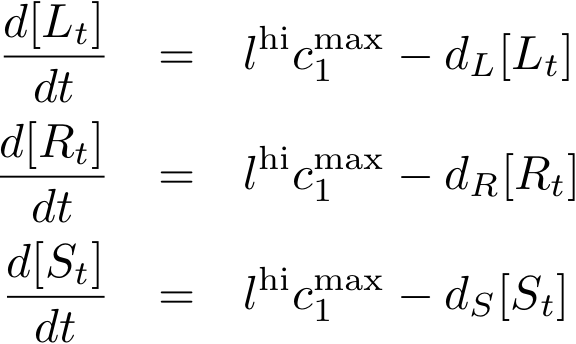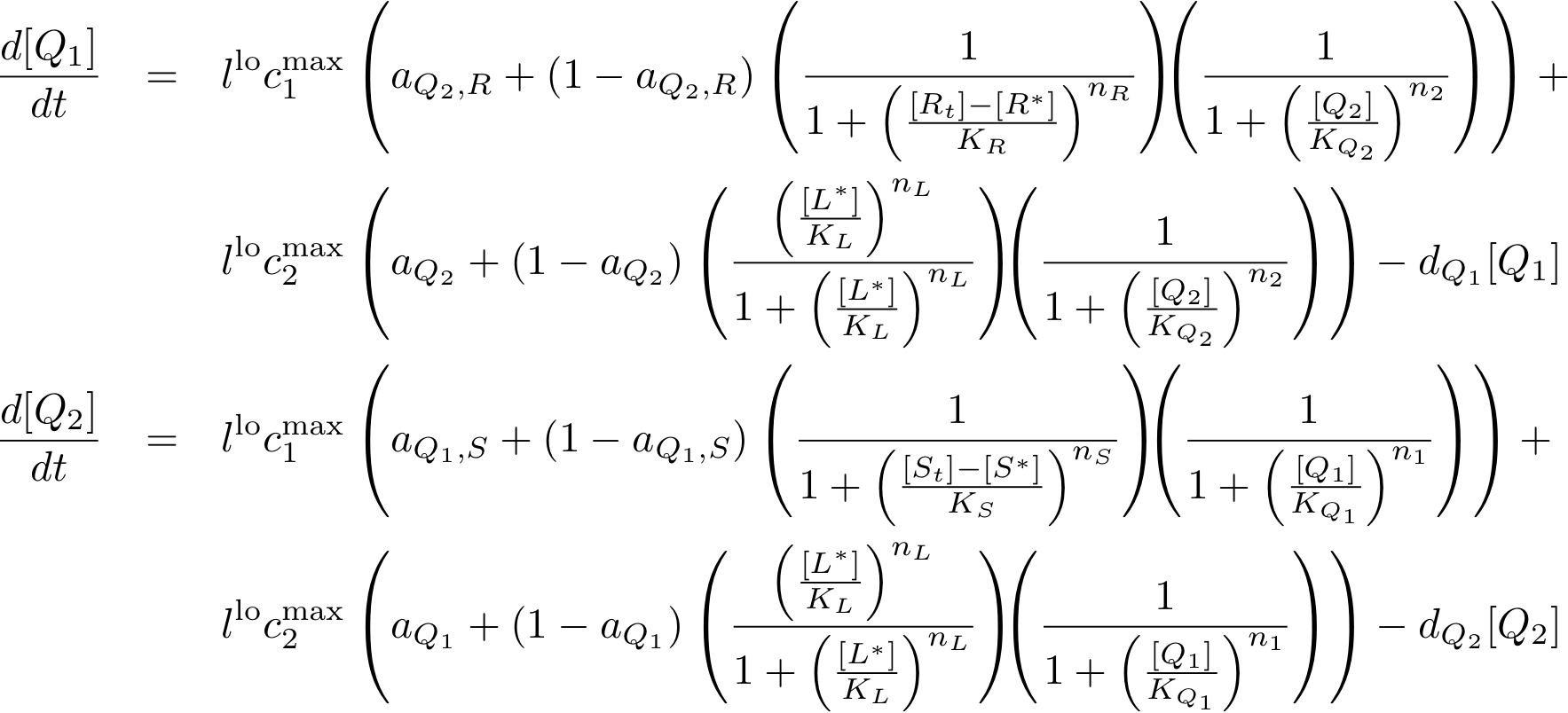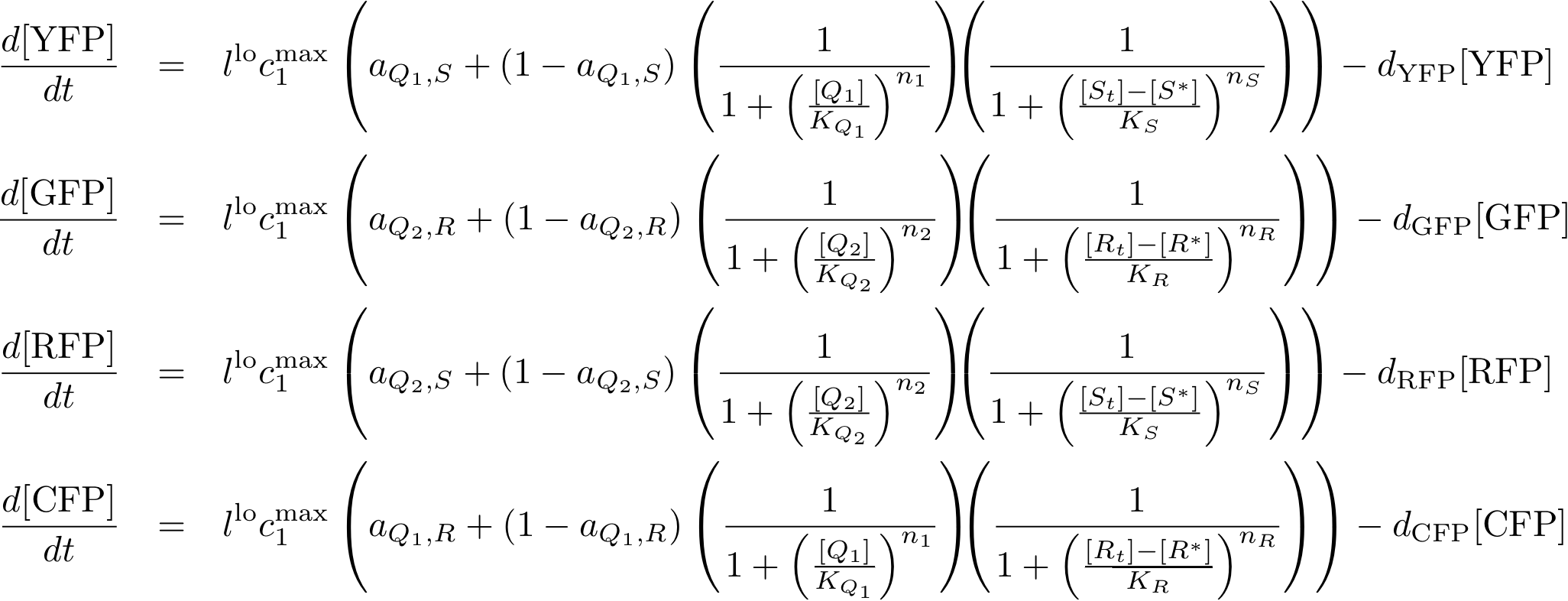ETHZ/Simulations
From 2007.igem.org
Contents |
Basic Model
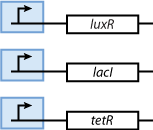
Constitutively produced proteins
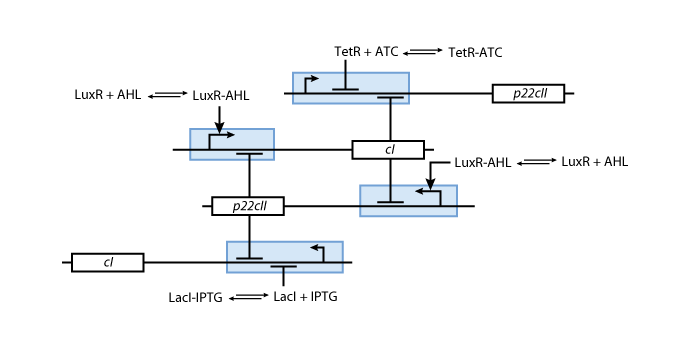
Learning system
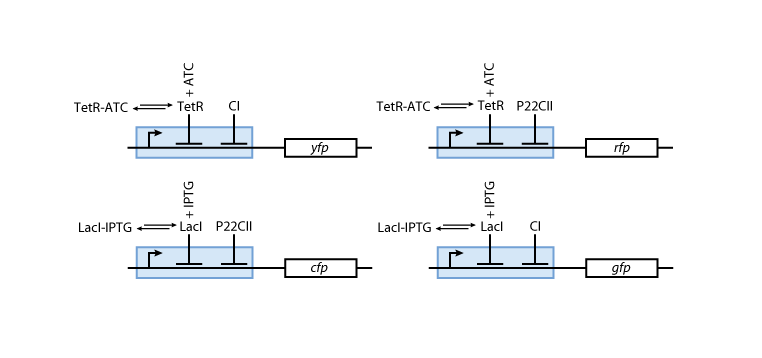
Reporter system
System Equations
Constitutively produced proteins
Learning system
Reporter system
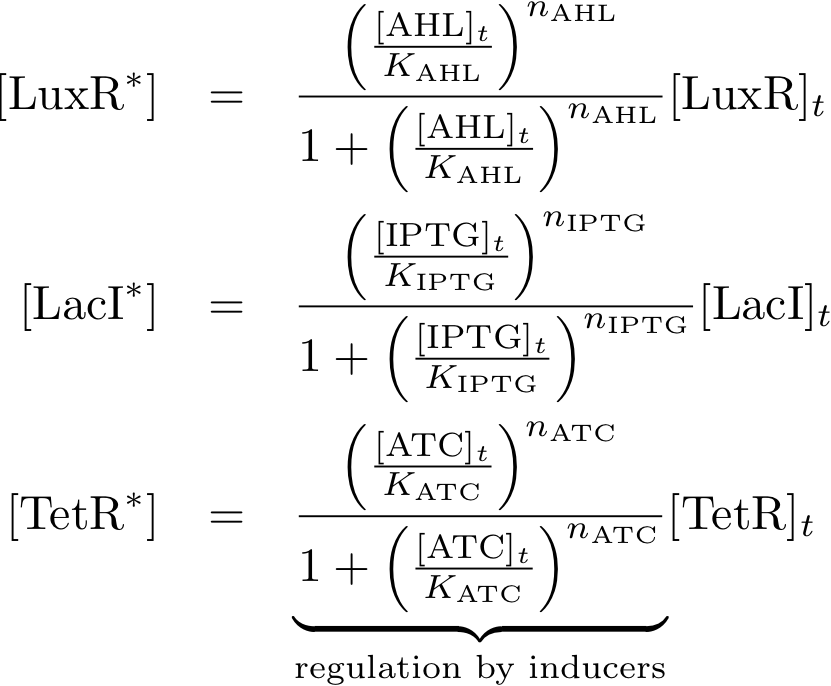
Allosteric regulation
Comments
Note that the three constitutively produced proteins lacI, tetR and luxR exist in two different forms: as free proteins and in complexes they build with IPTG, aTc and AHL, respectively.
In this new formulation of the model equations, the characterization is more amenable to human interpretation (although equivalent to the previous formuation). The promoters are now characterized by their maximum transcription rate (cimax) and the basic production (aX), which gives the 'leakage' if the gene is fully inhibited. Note that in the given mathematical formulation the basic production is specified as a percentage of the max. transcription rate and is therefore unitless.
The max. transcription rate is given per gene (as agreed with Sven during the meeting at Sep 20.). This means that to get the total transcription rate we need to multiply with the number of gene copies per cell which is represented as llo/lhi in the model equations.
Model Parameters
| Parameter | Value | Description | Comments |
|---|---|---|---|
| c1max | 0.01 [mM/h] | max. transcription rate of constitutive promoter (per gene) | promoter no. J23105; Reference: Estimate |
| c2max | 0.01 [mM/h] | max. transcription rate of luxR-activated promoter (per gene) | Reference: Estimate |
| lhi | 25 | high-copy plasmid number | Reference: Estimate |
| llo | 5 | low-copy plasmid number | Reference: Estimate |
| aQ2,R | 0.1 - 0.2 | basic production of Q2/R-inhibited genes | Reference: Conclusions after discussion |
| aQ2 | 0.1 - 0.2 | basic production of Q2-inhibited genes | Reference: Conclusions after discussion |
| aQ1,S | 0.1 - 0.2 | basic production of Q1/S-inhibited genes | Reference: Conclusions after discussion |
| aQ1 | 0.1 - 0.2 | basic production of Q1-inhibited genes | Reference: Conclusions after discussion |
| aQ2,S | 0.1 - 0.2 | basic production of Q2/S-inhibited genes | Reference: Conclusions after discussion |
| aQ1,R | 0.1 - 0.2 | basic production of Q1/R-inhibited genes | Reference: Conclusions after discussion |
| dR | 2.31e-3 [per sec] | degradation of lacI | Ref. [10] |
| dS | 1e-5 [pro sec]/2.31e-3 [per sec] | degradation of tetR | Ref. [9]/ Ref. [10] |
| dL | 1e-3 - 1e-4 [per sec] | degradation of luxR | Ref: [6] |
| dQ1 | 7e-4 [per sec] | degradation of cI | Ref. [7] |
| dQ2 | degradation of p22cII | ||
| dYFP | 6.3e-3 [per min] | degradation of YFP | suppl. mat. to Ref. [8] corresponding to a half life of 110min |
| dGFP | 6.3e-3 [per min] | degradation of GFP | in analogy to YFP |
| dRFP | 6.3e-3 [per min] | degradation of RFP | in analogy to YFP |
| dCFP | 6.3e-3 [per min] | degradation of CFP | in analogy to YFP |
| KR | 0.1 - 1 [pM] | lacI repressor dissociation constant | Ref. [2] |
| KIR | 1.3 [µM] | IPTG-lacI repressor dissociation constant | Ref. [2] |
| KS | 179 [pM] | tetR repressor dissociation constant | Ref. [1] |
| KIS | 893 [pM] | aTc-tetR repressor dissociation constant | Ref. [1] |
| KL | 55 - 520 [nM] | luxR activator dissociation constant | Ref: [6] |
| KIL | 0.09 - 1 [µM] | AHL-luxR activator dissociation constant | Ref: [6] |
| KQ1 |
| cI repressor dissociation constant |
|
| KQ2 | 0.577 [µM] | p22cII repressor dissociation constant | Ref. [11]. Note that they use a protein cII and we have p22cII. Does that match? |
| nR | 1 | lacI repressor Hill cooperativity | Ref. [5] |
| nIR | 2 | IPTG-lacI repressor Hill cooperativity | Ref. [5] |
| nS | 3 | tetR repressor Hill cooperativity | Ref. [3] |
| nIS | 2 (1.5-2.5) | aTc-tetR repressor Hill cooperativity | Ref. [3] |
| nL | 2 | luxR activator Hill cooperativity | Ref: [6] |
| nIL | 1 | AHL-luxR activator Hill cooperativity | Ref. [3] |
| nQ1 | 2 | cI repressor Hill cooperativity | Ref. [12] |
| nQ2 | 4 | p22cII repressor Hill cooperativity | Ref. [11]. Note that they use a protein cII and we have p22cII. Does that match? |
References
[http://www.pnas.org/cgi/content/abstract/104/8/2643 [1] Weber et al. (2007) P Natl Acad Sci USA 104(8):2643-2648]
[http://www.pnas.org/cgi/content/full/100/13/7702?ck=nck [2] Setty et al (2003) P Natl Acad Sci USA 100(13): 7702-7707]
[http://ieeexplore.ieee.org/iel5/9711/30654/01416417.pdf [3] Braun et al (2005) ICASSP 5:769-772]
[http:// [4] Supplementary on-line information for "A Synthetic gene-metabolic oscillator"]
[http://dx.doi.org/10.1016/j.jbiotec.2005.08.030 [5] Ladevaia and Mantzais (2006) J Biotechnol 122(1):99-121]
[http://dx.doi.org/10.1016/j.biosystems.2005.04.006 [6] Goryachev et al. (2004) Biosystems 83(2-3):178-187]
[http:// [7] Stochastic Kinetic Analysis of Developmental Pathway Bifurcation in Phage λ-Infected E. coli Cells]
[http:// [8] Yeast Cbk1 and Mob2 Activate Daughter-Specific Genetic Programs to Induce Asymmetric Cell Fates]
[http:// [9] Engineering stability in gene networks by autoregulation]
[http:// [10] Model-Driven Designs of an Oscillating Gene Network]
[http://www.pnas.org/cgi/reprint/99/2/679 [11] McMillen et al. (2002) P Natl Acad Sci USA 99(2):679-684]
[http://www.nature.com/nature/journal/v434/n7037/full/nature03461.html [12] Basu et al. (2005) Nature 434:1130-1134]
Variable Mapping
| Variable | Compound |
|---|---|
| R | lacI |
| IR | IPTG |
| S | tetR |
| IS | aTc |
| L | luxR |
| IL | AHL |
| Q1 | cI |
| Q2 | p22cII |