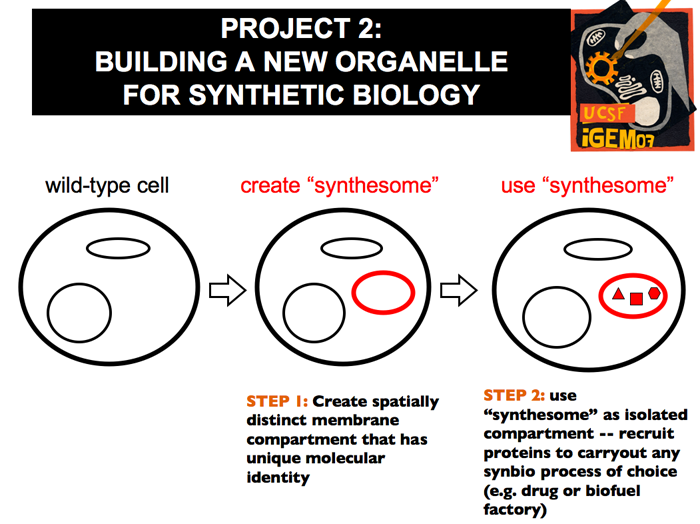
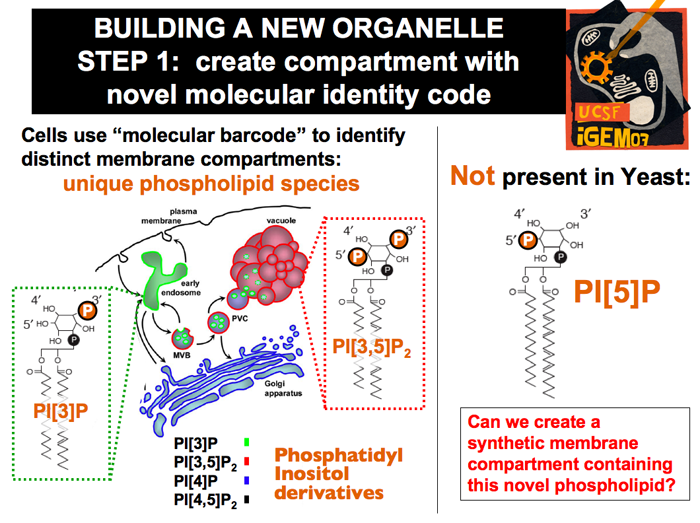
 | The existence of different organelles bound by distinct membranes necessitates a system by which the cell can distinguish different membrane locations. One of the most well-understood methods of tagging such membranes is the phosphoinositide system. Phosphoinositides are lipids that make up a small percentage of membranes. They consist of a hydrophobic tail and an inositol sugar head that can be phosphorlyated in 3 positions. In yeast, there are no insositides tagged solely at 5’ position.
|
|
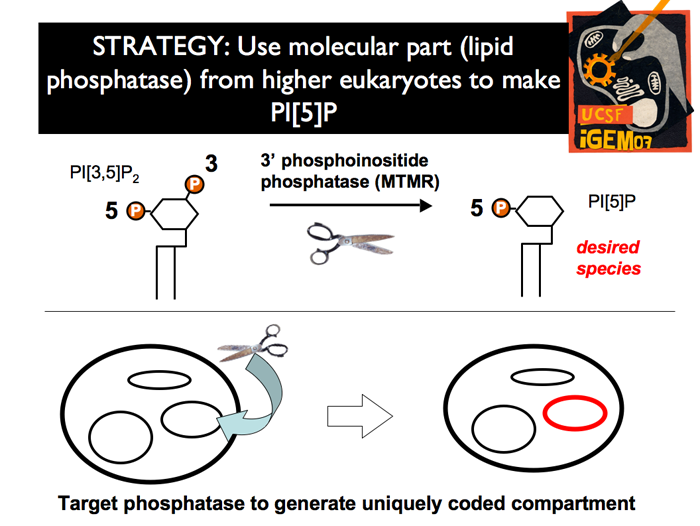 | Because of this, if we created this new tag in a specific location, we would theoretically obtain a modified vesicle to serve as a chassis for subsequent recruitment of other proteins, which through modular domains able to bind only to the desired phosphoinositide.
We have chosen the following strategy: express in yeast the higher eukaryote lipid phosphatase MTM, which catalyzes the removal of the 3-phosphate from PI[3,5]P2, to generate PI[5]P in a specific location.
|
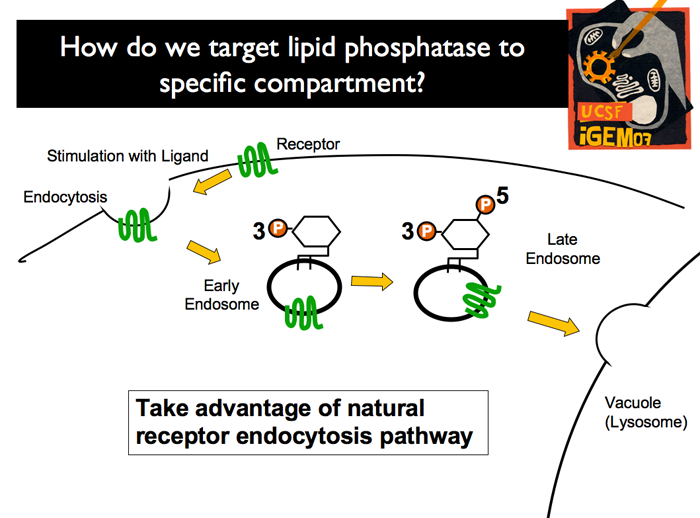 | The endogenous system in yeast has a membrane associated mating-pathway receptor wchih binds a ligand (mating pheromone). After ligand binding, the receptor-phermone complex is endocytosed. After endocyctosis, an endogenous kinase phosphorylates the yet untagged phosphoinositide on the 3’ position. As the early endosome is absorbed into the late endosome, a second endogenous kinase phosphorylates the 5’ position, resulting in a PI[3,5]P2 tagged endosome. This endosome is recognized by the vacuole, and is then targeted by its merger machinery for lysis.
|
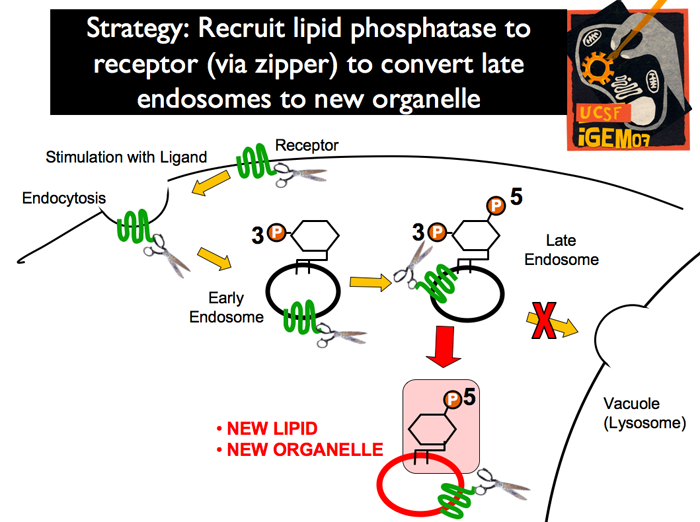 | In our strategy, human or worm MTM will be recruited to the membrane receptor of the mating pathway (Ste2). Pathway induction with mating pheromone (alpha factor) results in endocytosis of the receptor (and therefore the recruited MTM). Once in the late endosome, the MTM will convert from PI[3,5]P2 into PI[5]P, leading to Ste2-specific PI[5]P-tagged endosomes. These endosomes are expected to be stable, for they are unlikely to be recognized by the vacuole merging machinery.
|