Talk:Tokyo/Assays for hybrid promoter
From 2007.igem.org
Contents |
AHL assay for Lux-lac hybrid promoter
Purpose:
check if AHL activates lux-lac hybrid promoter
check if lacI represses lux-lac hybrid promoter
Samples:
hybrid promoter in pTrc99A
hybrod promoter in pBR322
luxR in pTrc99A
luxR --- AHL-dependent activation confirmed
placqi on promter-less GFP in DH5a (for pos. con.)
promoter-less GFP in DH5a (for neg. con.)
Procedure:
prepare overnight culture for each sample
make fresh culture
take 3 ul of the overinight culture into 3 ml of LB (Amp and/or Kan) in Falcon tubes.
incubate for 2 to 3 hours until the observed OD is around 1.2 (Falcon tube = 14 mm in daimeter)
add AHL & IPTG solution
[AHL]final (in 3 ml LB culture) = 10 nM
[IPTG]final (in 3 ml LB culture) = 1 mM
incubate for 2 to 3 hours
apply 150 ul of samples into 96-well plaste
FLA measurement
Result:
Conclusion:
AND gate by AHL & IPTG
Lux-lac hybrid promoter is activated only in the presence of AHL and IPTG.
IPTG assay
Purpose:
To determine the order of the concentration of IPTG necessary for the activation of our lux-lac hybrid promoter in the LacI producing pTrc99A cells.
The order of the concentration is used for more detailed assay with narrower range of the IPTG concentration.
Samples:
A4 placQI in pTrc99A
A4 ΔP in pTrc99A
Lux-lac hybrid promter + A4 in pBR322
Lux-lac hybrid promter + A4 in pTrc99A
Procedure:
prepare overnight culture for each sample
make fresh culture
take 3 ul of the overinight culture into 3 ml of LB (Amp and/or Kan) in Falcon tubes.
incubate for 2 to 3 hours until the observed OD is around 0.5
add AHL & IPTG solution
[AHL]final (in 3 ml LB culture) = 10 nM
[IPTG]final (in 3 ml LB culture) = 1000, 100, 10, 1, 0.1, 0.01, and 0 mM
incubate for 2 to 3 hours
apply 150 ul of samples into 96-well plaste
FLA measurement
Result:
Conclusion:
AHL assay – Lux-lac hybrid promoter activation by endogenous AHL
Purpose
To check if worker E. coli (Sender) can produce enough AHL for our model to work by using different copy numbers of plasmids.
Samples
placI luxI on PSB1(high copy) and A4Δp(Sender 1)
placI luxI on PSB4(low copy) and A4Δp(Sender 2)
placI luxI on PBR322(low copy) and A4Δp(Sender 3)
(no promoter)tetR pBR322 and A4 Hybrid promoter(Receiver 1)
(no promoter)luxI pBR322 and A4Δp
Procedure
0:00 Start to prepare over night culture (1 tube for each)
12:00 Make fresh culture with 3 ul of the overnight culture in 3ml of LB + Amp + Kan (LBAK) (3 tubes for each)
Incubation in a shaker until the observed OD (ODobs) reaches up to 0.2
Equalize ODobs of all the samples by adding LB media.
15:00 Centrifugation at 5000 rpm and wash out the LB media.
Add cells with A4Δp and each of the following to 2.4 ml of LBAK
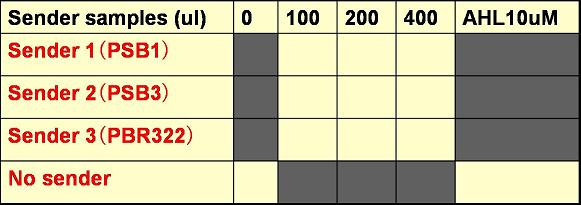
The experiments tested yellow boxes, not gray ones.
The total volume of E. coli culture (A4Δp and senders) was always 400 uM.
Wash
16:00 (incubate it until the OD obs. <0.20)
17:30 stop incubation => wash* => FLA analysis(When OD reached 0.8)
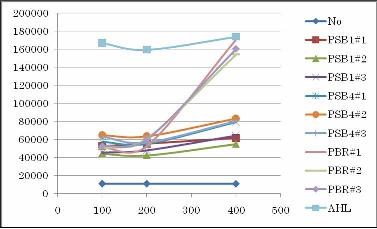
Results
Conclusion
Not only high copy number plasmid pSB1, also low copy number plasmid pSB4 and pBR produced enough AHL to activate the LacI hybrid promoter in other cells. Especially, pBR remarkably produced AHL in the present experiment.