Bologna University/Image Acquisition and Elaboration
From 2007.igem.org
Image Acquisition
Using the experimental set up illustrated here, we obtain short film segments of our fluorescent bacteria; then, with an apposite software, we extract several frames from each of them. Examples of images we get are shown below.
Image Elaboration
Since we need to know how much of the total image area is occupied by bacteria, we process the images with our segmentation algorithm implemented in Matlab.
Here are the processing phases:
- the RGB image is read and the green channel is extracted;
- a morphological top hat filtering is performed on the grayscale image;
- by means of an adaptive threshold, the image is tranformed in a binary one;
- the distance between every point and the nearest black pixel is calculated;
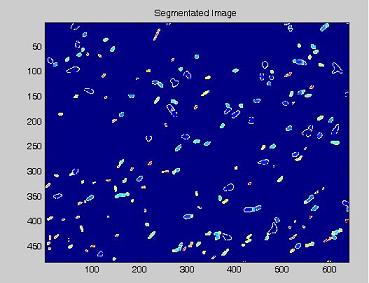
- on this last image the [http://en.wikipedia.org/wiki/Watershed_%28algorithm%29 watershed] algorithm is applied: every pixel is assigned a label, depending on the segmentated region it belongs; then, every labeled region is represented with a different colour, as in the images shown below;
- the area of every segmented region is calculated, checking that the summation of these areas (including the background) balances the image total area;
- two complementary matrices are created:
-ImageWithOutBackground, containing the intensity positive values corresponding only to pixels recognised as bacteria, with zeros elsewhere; -ImageBackground, which contains the intensity positive values of pixels recognised as background, with zeros as other entries;
- with a summation over all the ImageWithOutBackground matrix' entries, the total intensity is obtained;
- dividing this value by the total bacteria area, the output is the normalized intensity we use to compare the fluorescence of different kinds of bacteria.
Measures to validate acquisition and elaboration protocol
We use bacteria with [http://partsregistry.org/Part:BBa_I763004 I763004] plasmid, since we know all of them beam fluorescence also when not inducted; we check that the OD value is 1.2. We then execute two different observations:
- ten different spots within the same slide (test A);
- five slides, and two spots each (10 total spots, test B).
For every spot, we take a picture and we elaborate it using the algorithm above, obtaining as output a normalized intensity value. Finally, for each of the two sets of values, we calculate mean and standard deviation; for the B test, we calculate both values either on the 10 intensities indistinctly (B1) and on the five slide means (B2). Here are the results:
| Mean | Std | |
|
|
|
As we expected, the two mean values are close to each other, being the different spots from the same solution. The standard deviation is small enough for our measures to be considered well-founded and our protocol validated.
Fluorescence reliance on Promoter and Medium
We test bacteria with two different plasmids, [http://partsregistry.org/Part:BBa_I763004 I763004] and [http://partsregistry.org/Part:BBa_I763035 I763035], each of them into two different mediums, LB and M9, to evaluate the dependence of the output fluorescence intensity on both the promoter and the medium. The experimental setup conditions are the same for each test, and the OD value is mantained at 1.2. We take pictures from two spots on each of the five slides we prepare; then, we calculate the mean value for each slide and finally the mean and the standard devioation on these values. Here are the results:
| LB | M9 | |||
|---|---|---|---|---|
| I763004 | 102,852 | 13,39113684 | 123,2375 | 13,06524493 |
| I763035 | 106,575 | 6,040488391 | 138,0875 | 7,707288217 |