Imperial/Infector Detector/Conclusion
From 2007.igem.org

Infector Detector: Conclusion
The main achievements of the Infector Detector project:
- Extensive modelling of the two potential constructs for Infector Detector
- Purification of GFPmut3b to allow construction of a calibration curve
- Detailed characterisation of construct 1 in vitro using a calibration curve to find rate of GFP synthesis
- Creation of a standard unit to allow comparison between in vitro and in vivo
The table below summarises our Infector Detector system in the context of the original specifications:
| Achievements | ||
| Inputs | Sensitive to 5-1000nM | |
| Outputs | Future work - Using Stronger fluorescent protein such as DsRed express | |
| Response Time | Systems responds <30minutes | |
| Operating Conditions | System works at 25°C | |
| Health & Safety | Cell Free in vitro chassis | |
| Shelf-life | Can be stored in freezer for prolonged periods | |
| Packaging | Future Work - Using our chassis in a spray |
Battle a spectrum of infections
The great potential of Infector Detector is that it is not limited to just one type of infection. Adding sensitivity to AHL originating from biofilms is just the beginning. By tweaking the internal mechanisms of the construct, Infector Detector can be used to battle a range of catheter-related bacteremias. By using a construct that recognises AI-21, for example, we can detect the presence of Klebsiella pneumoniae, a pathogenic bacterium ranked second to E. coli for urinary tract infections in older persons.
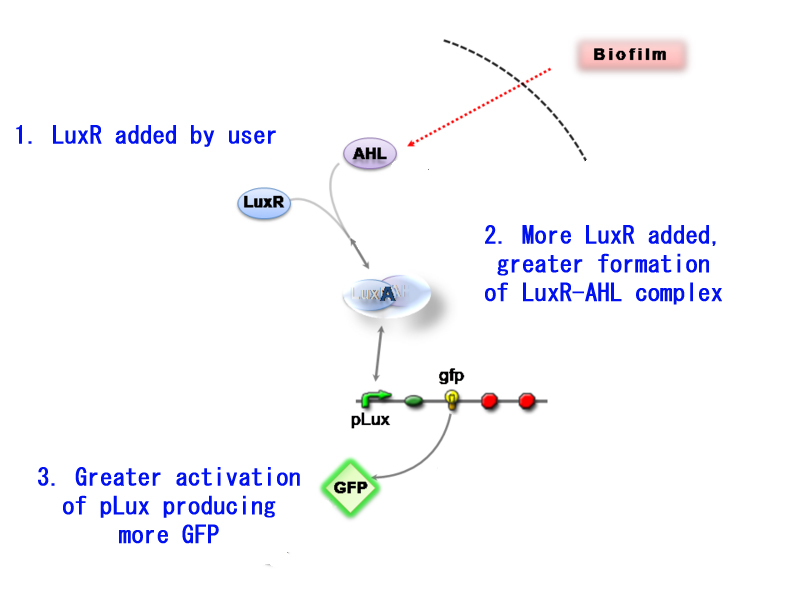
Added control - Construct 2
The main advantage of using construct 2 is that it provides an additional control mechanism for our detector meaning that you can tweak the detector sensitivity.
Going into deeper detail, construct 1 can produce LuxR as soon as it is activated. LuxR's presence is necessary for the formation of AHL-LuxR complex and the subsequent activation of pLux (leading to GFP production). Construct 2 on the other hand does not have a LuxR producing part. It relies on the user to add the necessary LuxR to form the binding complex. This control over LuxR can thus act as a sort of attenuator to the sensitivity of Infector Detector.
Having little LuxR present, will form very little binding complex with AHL and thus the sensitivity will decrease significantly. Saturating the detection compound with LuxR will maximise the sensitivity. Briefly, if we want to detect only highly progressed infections, we add little LuxR. If we want to detect infections with minimum progression, we saturate with AHL.
Thus construct 2 can be used as a sensitivity attenuator.
Packaging
Infector Detector can be packaged as either a cream or a spray.
A spray will provide easy application of the detector because it does not require the user to fiddle around with the urinary catheter as he can simply spray from a distance. The disadvantage being poor accuracy of application and more evaporation.
A cream on the other hand will decrease significanlty any evaporation and will allow the user to apply the detector to specific areas of the catheter without the possibility of spraying the patient itself with detector. The disadvantage being here is the low diffusion rates of AHL and other compounds that need to cross the viscous cream to reach the actual detector assemblies. This might hinder rapid detection.
Both applications provide some advantages and disadvantages that must be weighed depending on the actual use scenario of Infector Detector in order to decide which is best.
References
- Damien Balestrino et al. Characterization of Type 2 Quorum Sensing in Klebsiella pneumoniae and Relationship with Biofilm Formation. J Bacteriol. 2005 April; 187(8): 2870–2880.