Dry to Wet
From 2007.igem.org
(→Application) |
(→Michaelis-Menton) |
||
| Line 22: | Line 22: | ||
=== Michaelis-Menton === | === Michaelis-Menton === | ||
| - | Anybody who has done any sort of biological study will know michaelis menton what i am trying to acheive here is to take it from the basics so as to equate it to the equations we will be using to model the system and to give the biologists an idea of what values and models we need. | + | ''This was taken from 'Biochemistry' by 'Straya''<br> |
| + | Anybody who has done any sort of biological study will know michaelis menton what i am trying to acheive here is to take it from the basics so as to equate it to the equations we will be using to model the system and to give the biologists an idea of what values and models we need. In this case all k values are rate constants and [] means concentration and E is enzyme, S is substrate and [E]t is total enzyme concentration. <br> | ||
The michaelis-Menton equation describes the kinetic properties of many enzymes. Consider the simple system A -> P | The michaelis-Menton equation describes the kinetic properties of many enzymes. Consider the simple system A -> P | ||
The rate of V is the quantity of A that disappears over a specified unit of time which is equal to the rate of appearance of P. For this system V=k[A] where k is the rate constant. | The rate of V is the quantity of A that disappears over a specified unit of time which is equal to the rate of appearance of P. For this system V=k[A] where k is the rate constant. | ||
| - | The simplest model that accounts for the kinetic properties of many enzymes is | + | The simplest model that accounts for the kinetic properties of many enzymes is (i will add it in when i have figured out how to)<br> |
| + | what we want is an expression that relates the rate of catalysis to the concentrations of substrate and enzyme and the rates of the individual steps.<br> | ||
| + | Our starting point is that the catalytic rate is equal to the product of the ES complex and k3. | ||
| + | '''Vo=K2[ES]''' ''call this '''eqn(1)''' as i will be referring to it again'' | ||
| + | Now expressing [ES]in terms of known quantities the rates of '''formation''' and '''breakdown''' of [ES] are given by: | ||
| + | '''formation [ES] = k1*[E]*[S]''' | ||
| + | '''breakdown [ES] = (k2+k3)*[ES]''' | ||
| + | A steady state occurs when the rates of formation and breakdown of the ES complex are equal, this gives the formula | ||
| + | '''k1*[E]*[S]=(k2+k3)*[ES]''' | ||
| + | which then gives | ||
| + | '''[E][S] / [ES]=(k2+k3) / k1''' <br> | ||
| + | This can be simplified by defining '''Km''' called the michaelis constant<br> | ||
| + | '''Km = (k2+k3) / k1''' | ||
| + | from this we get | ||
| + | '''[ES] = [E][S] / Km''' ''call this '''eqn(2)''' as i will be referring to it again'' <br> | ||
| + | Now examining the numerator of this equation: because substrate is usually present at much higher concentrations than the enzyme, the concentration of uncombines substrate [S]is very nearly equal to the total substrate concentration. The concentration of enzyme [E] is equal to '''[E]t - [ES]''' now substituting this into ''eqn(2)'' and after some simplification we get | ||
| + | '''[ES]=([E]t*[S]) / ([S]+Km)''' | ||
| + | by substituting this expression into ''eqn(1)'' we get | ||
| + | '''Vo = (k2[E]t*[S]) / ([s]+Km)''' | ||
| + | The maximised rate Vmax is obtained when the catalytic sites on the enzyme are saturated with substrate i.e '''[ES] = [E]t''' thus | ||
| + | '''Vmax = k2*[E]t | ||
| + | this gives the Michaelis Menton equation | ||
| + | '''Vo = Vmax*([S] / ([S]+km)) ''' ''call this '''eqn(3)''' as i will refer to it again'' | ||
| + | when '''[S]=Km''' then '''Vo = Vmax / 2'''. Thus Km is equal to the substrate concentration at which the reaction rate is half its maximal value.<br> | ||
=== Sum & And Promoters === | === Sum & And Promoters === | ||
Revision as of 15:02, 11 July 2007
Contents |
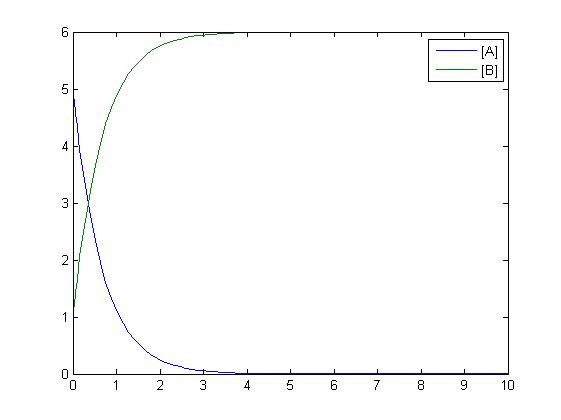
Mass-Action Reaction Modelling
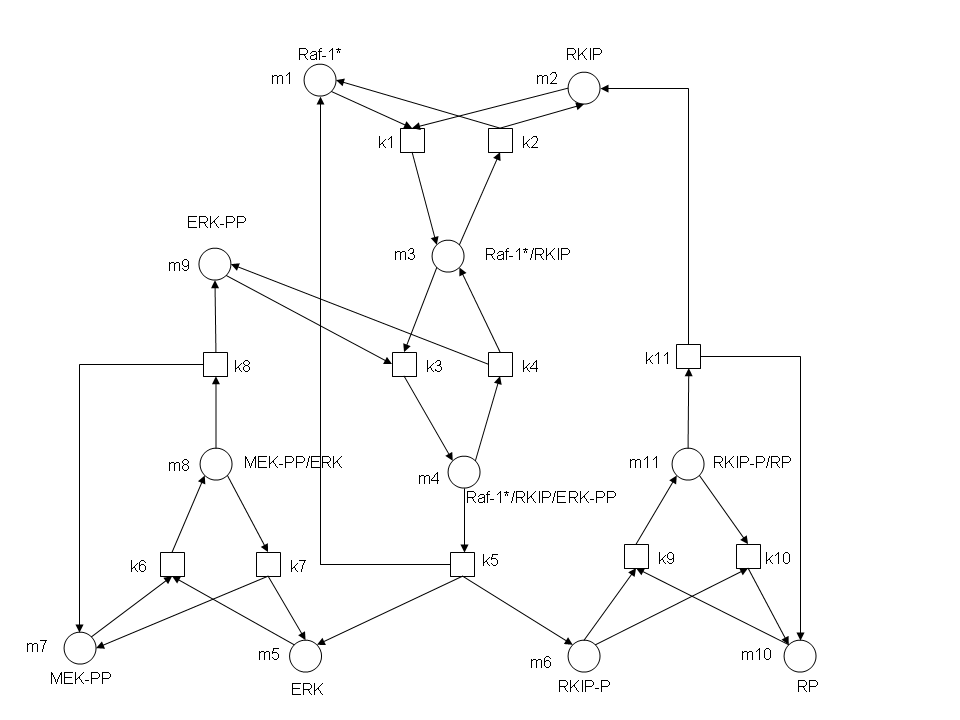
RKIP network
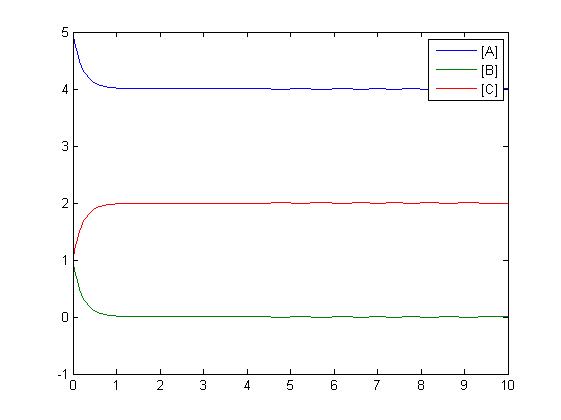
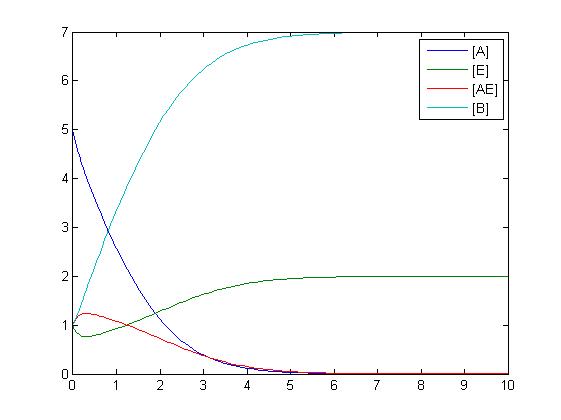
After gaining a thorough understanding of methods involved with modeling simple mass-action reactions, we can move on to more complex systems such as the RKIP network.
In the above diagram, substrates, enzymes and substrate/enzyme complexes are represented by numbered circles, rate constants are represented by numbered squares. By isolating individual species and their direct peripheral species (those being formed from or forming the isolated species) we are able to treat the group as a simple mass-action reaction. A differential equation is then found for each species based on the rate constants and code can be written and a graph plotted showing the trend of all the species’ concentration over time giving the following graph:
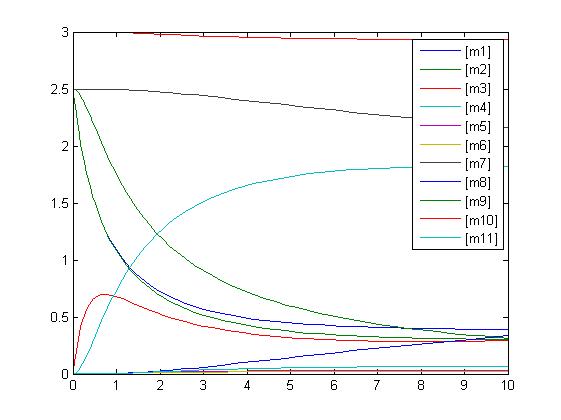
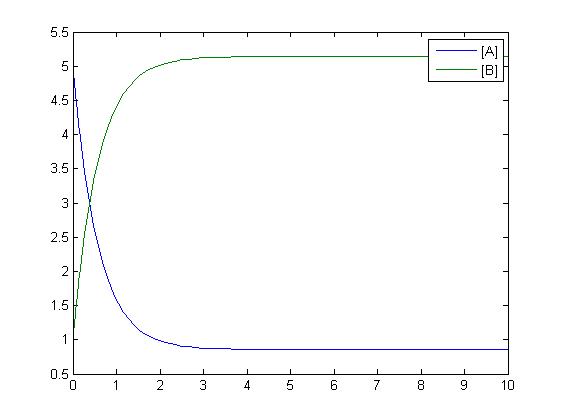
Sensitivity
An insight into a system's sensitivity will show how the variation of a model can be apportioned qualitatively or quantitatively to different sources of variation
One method of exposing the variation of a model is to program a loop exposing a modelled reaction to increasing values of a chosen constant. This process was followed with the metabolic pathway showing in Mass-Action Reaction Modelling and ploted on a graph showing the response of all 4 species for a set range of varying K2 values from 1 to 10 where 10 is highlighted red.
Michaelis-Menton
This was taken from 'Biochemistry' by 'Straya
Anybody who has done any sort of biological study will know michaelis menton what i am trying to acheive here is to take it from the basics so as to equate it to the equations we will be using to model the system and to give the biologists an idea of what values and models we need. In this case all k values are rate constants and [] means concentration and E is enzyme, S is substrate and [E]t is total enzyme concentration.
The michaelis-Menton equation describes the kinetic properties of many enzymes. Consider the simple system A -> P
The rate of V is the quantity of A that disappears over a specified unit of time which is equal to the rate of appearance of P. For this system V=k[A] where k is the rate constant.
The simplest model that accounts for the kinetic properties of many enzymes is (i will add it in when i have figured out how to)
what we want is an expression that relates the rate of catalysis to the concentrations of substrate and enzyme and the rates of the individual steps.
Our starting point is that the catalytic rate is equal to the product of the ES complex and k3.
Vo=K2[ES] call this eqn(1) as i will be referring to it again
Now expressing [ES]in terms of known quantities the rates of formation and breakdown of [ES] are given by:
formation [ES] = k1*[E]*[S] breakdown [ES] = (k2+k3)*[ES]
A steady state occurs when the rates of formation and breakdown of the ES complex are equal, this gives the formula
k1*[E]*[S]=(k2+k3)*[ES]
which then gives
[E][S] / [ES]=(k2+k3) / k1
This can be simplified by defining Km called the michaelis constant
Km = (k2+k3) / k1
from this we get
[ES] = [E][S] / Km call this eqn(2) as i will be referring to it again
Now examining the numerator of this equation: because substrate is usually present at much higher concentrations than the enzyme, the concentration of uncombines substrate [S]is very nearly equal to the total substrate concentration. The concentration of enzyme [E] is equal to [E]t - [ES] now substituting this into eqn(2) and after some simplification we get
[ES]=([E]t*[S]) / ([S]+Km)
by substituting this expression into eqn(1) we get
Vo = (k2[E]t*[S]) / ([s]+Km)
The maximised rate Vmax is obtained when the catalytic sites on the enzyme are saturated with substrate i.e [ES] = [E]t thus
Vmax = k2*[E]t
this gives the Michaelis Menton equation
Vo = Vmax*([S] / ([S]+km)) call this eqn(3) as i will refer to it again
when [S]=Km then Vo = Vmax / 2. Thus Km is equal to the substrate concentration at which the reaction rate is half its maximal value.
Sum & And Promoters
Application
<math>x</math>