Samantha Liang Notebook
From 2007.igem.org
Current Notebook July 20, 2007 to present
My Construction Files
My Sequencing Files
My Biobricks
My -80 Stocks
Things to read about:
- Does barnase have DNase activity?
- Is there a way to do dapi (pronounced dap-E) measurements for e.coli? dapI binds to DNA, wonder if can use a cytometer or something? Do a dapi stain?
- Make sure that we're not just making a toxic protein or that Cre itself is toxic or something like that.
For tomorrow:
- Miniprep 408C, 408E, and 411B
- Sequence 408C, 408E, and 411B
- Make -80s of 408C, 408E, and 411B
- Pool I716430 through I716435
- Look at spotted plates
Samanthaliang 18:53, 19 July 2007 (EDT)
- Analyze sequencing results
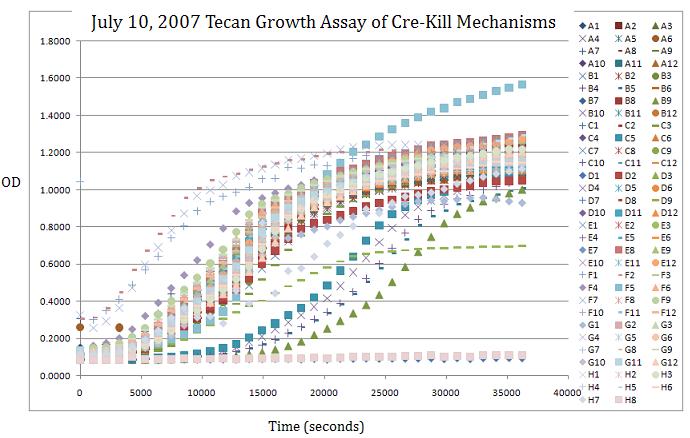
- Analyze Tecan results
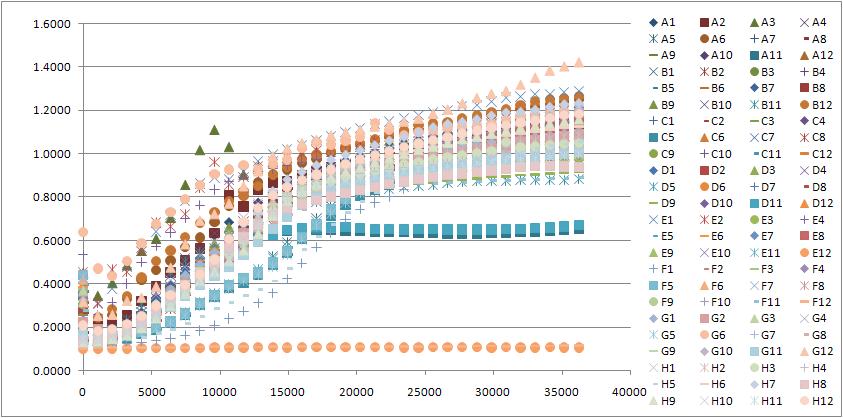
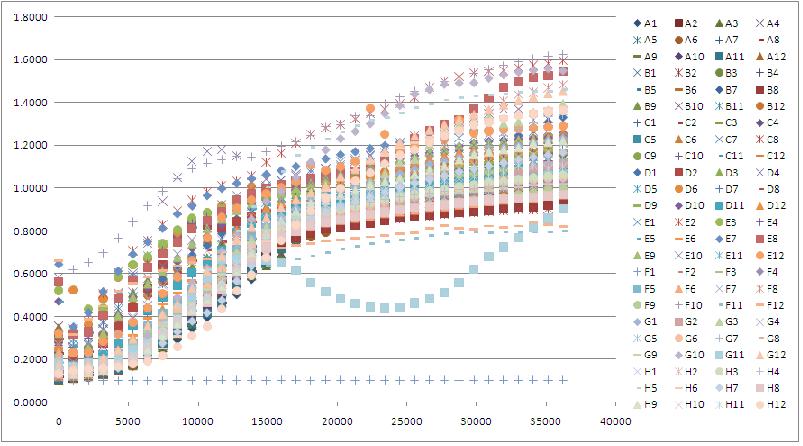
Note: There are a lot of graphs that go with this Tecan run, but maybe I'll post a few tomorrow. Basically 411B (G.2.bglII) didn't grow whether it was induced or not induced but this could be good becuase by putting it on a low copy plasmid, it may grow better when not induced. We are looking for a strong phenotype so absolutely no growth is ideal. However 408C and 408E show some promise too (G.2.barnase) because when not triggered they grow at the same rate as the control DH10B, but when triggered, they plateau at almost half the OD, meaning that they should be dying.
- Grow up 408C and 408E cultures by transferring 1ul from the 96 well blocks to a test tube of 5ml
- Try to grow up a 411B colony in liquid media by adding some glucose, which will block some of the pBad activity
- Pick a colony and swirl it in 100ul of LB, then smear it all over a plate - can miniprep straight from the plate tomorrow
- Spot arabinose plates with tecan wells to see if they grow (pay special attention to 408C and 408E)
- Miniprep I716424 through I716429
- Subclone I716430 through I716435 by replacing the CamR of I716424 through I716429 with pBad
Samanthaliang 19:56, 18 July 2007 (EDT)
- Analyze Tecan results and look at streaked plates to make sure those clones are no longer viable - all are viable on LB-carb plates unfortunately (spot on arabinose plates next time)
- Save retransformed 401, 410, 407, 409, 416, 417, 418, 419, 420, 421 pools in fridge
- Pool I716424 through I716429
- Grow up cultures from the 96 well block for another Tecan growth assay
- Come back at 9:00pm today to put in the Tecan assay
Samanthaliang 15:20, 17 July 2007 (EDT)
- Miniprep 422 and 423 clones
- Miniprep Bca1109 (one for Chris, one for me)
- Miniprep 401, 410, 407, 409, 416, 417, 418, 419, 420, 421 pools
- Retransform 401, 410, 407, 409, 416, 417, 418, 419, 420, 421 pools
- Make -80s of 401, 410, 407, 409, 416, 417, 418, 419, 420, 421 pools
- Sequence 401, 410, 407, 409, 416, 417, 418, 419, 420, 421 pools
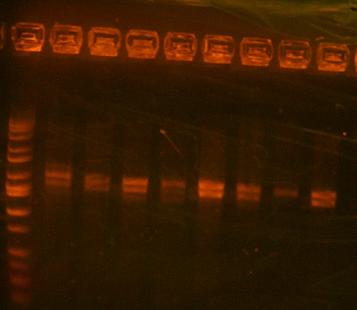
- Analytic digest of 422 and 423 clones
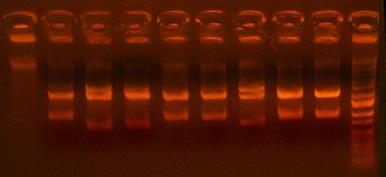
lanes 1-4 are I716422, lanes 5-8 are I716423, lane 9 is ladder
- Sequence 422 (clones 1&4)and 423 (clones 1&3) clones that look good
- Make -80s of 422 and 423 clones (clone 1 for both)
- Subclone [CamR][rbs][Cre][cassette][RFP] varaints (I716424 through I716429)
- Tecan growth assay
- Spot plates with cultures from the tecan wells that show a plateau characteristic
Notes on the Tecan growth assay today:
- Some wells in the 96 well block did not grow (E1, E3, E4, and E6) which are all 411Bs, G.2.BglIIs. This could mean that I just didn't pick colonies well, or else it is a pretty strong death switch which leaked a little early and died early. This could mean that the G.2.BglII has potential to work.
- Well H12 is blank, and G12 is control DH10B with arabinose
More Notes:
- In the future, do some tecan wells with arabinose and without arabinose to compare the different growth curves with and without an inducer
- Also, maybe try to lyse the cells in tecan wells that plateau in growth and run them on a gel to look at the DNA.
- What does it mean to destroy all the RNA with barnase? Look up if barnase has any DNase activity
- Ideally would we like to have a BglII or BamHI toxin because we could make an indestructible plasmid that destroys everything else.
- ceaB would be nice because it leaks out to other cells and kills them, but it may be too strong and hits may be falling out, which is why I'm not seeing results with it.
Samanthaliang 14:30, 16 July 2007 (EDT)
- Grow up colonies of I716422 and I716423
- Pool 401, 410, 407 and 409
- Grow up Bca1109
- Pool the 416-421 plates for backup (Next - Miniprep the 416 through 421 pools and retransform them, but let's see if there's better efficiency without a retransformation first)
- Autoclave 96 wells
- Grow up cultures for tecan growth assay (include isolated rbs+Cre combos + controls)
- All the restreaks worked except for the two A.1.barnases (407) which showed no growth on the tecan assays. On second look, I've realized that those wells did not grow to saturation, so it isn't the phenotype that we want
Samanthaliang 15:19, 13 July 2007 (EDT)
- Do Tecan assay for 412 through 421 with proper controls (11:00am-9:00pm after Weston)
- Analyzed yesterday's Tecan data - a 401 clone showed a suicide phenotype but since the sequencing came back for 401 as having a CamR marker instead of pBad, this may be from a pipetting error or something. Also, a couple of the 411 (G.2.BglII), 412(T.1.BglII) and 408 (G.2.barnase) showed a plateau of growth lower than an OD of 0.9500, while the OD of DH10B controls were 1.4507 and 1.3587, so these clones could be showing a suicide phenotype a little later than right away. Am going to restreak these cultures on plates so that it is no longer a library and repeat these growth experiments with multiple copies of the same thing and see if i can repeat the result. Can also grow some in wells with and without arabinose in order to get some good contrasts.
- Streak on plates from the 96 well block from 7-12-07:
- F1 (401) A.1.BamHI
- F11 (411) G.2.BglII
- F12 (407) A.1. barnase
- G11 (411) G.2.BglII
- C8 (408) G.2.barnase
- B8 (408) G.2.barnase
- Ask someone to put plates in the fridge
Later tonight:
- Look at Tecan data from today. Streak plates that look promising.
- Streaked plaes from 96 well block from 7-13-07:
- A11 (408) G.2.barnase
- B11 (408) G.2.barnase
- D9 (420) G.1.barnase
- D11 (408) G.2.barnase
- F11 (408) G.2.barnase
- F8 (419) T.1.barnase
Samanthaliang 19:33, 12 July 2007 (EDT)
- Grow up colonies in 96 well blocks of 412 through 421 (for spotted plate assay or Tecan growth assay) - grow to saturation
- Do Tecan assay for 401 through 411 with proper controls (10:00am-8:00pm after John and before Weston)
- Miniprep Bca1146 (RFP with a stop codon)
- Subclone cassette+RFP I716422 and I716423
- Make a painful amount of APE files
- Analyzed sequencing data - most of them came out with pBad in front which is good. However, 401 and 410 still had the CamR marker and 407 and 409 gave mixed reads
- Subclone 401, 410, 407 and 409 again
- Retransform Bca1109 (pBad promoter) because am almost out of the miniprep.
Note - If any cultures look promising on these first few tecans, you will need to start from the naive culture, miniprep, run more tests to make sure they are consistent. Also, because you won't be able to do restriction mapping to make sure the marker is excised since all the cells that have been excised will be dead, might want to induce some cultures for 3 hours or so, then dilute them, then spot them on plates with and without arabinose and get a percentage of how many die after so being induced for only 3 hours. Also might want to organize the data differently on charts, like group all the toxins together and see the effect of ATG start codons and compare the various toxin variants
Samanthaliang 14:34, 11 July 2007 (EDT)
- Grow up Bca1146 in DH10B
- Digest more pBad insert Bca1109 E/BamHI to insert into the suicide mechanisms I716416 through I716421
- Ligate and transform I716416 through I716421
- Sequence 401 through 415 to make sure pBad is there
- Sign up for Tecan Thursday and Friday
- Grow up cultures for better Tecan assay
- For controls include DH10B without arabinose, DH10B with arabinose)
- Save the naive cultures the next day in the fridge so that you can pick them
- This time you have 21 different possible toxin variants so do I716401 through I716411 of the variants today, and I716412 through I716415 tomorrow along with I716416 through I716421 if they grow.
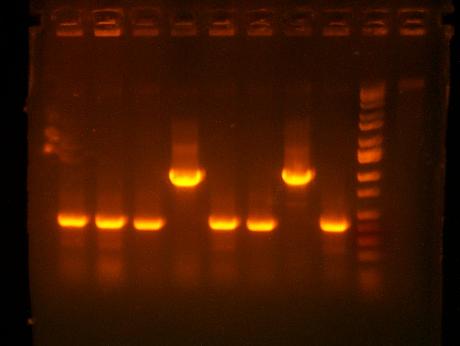
- Analyze growth curves from tecan assay
So I don't have the original naive cultures so I'll have to repeat this experiment but definitely D9 (I716408 - GTG Cre.cassette2dblterm.barnase) and D1 (I716407 - ATG Cre.cassette1.barnase) show some promise. It's nice to know that these constructs weren't a total failure and soon I'll have some ceaB and more barnase to try as well.
Samanthaliang 12:56, 10 July 2007 (EDT)
- Tecan growth assay
- Miniprep the pools
- Digest 228, 230, and 232, 241, 243, and 244 (EcoRI/BglII) to remove the CamR and replace it with pBad (Bca1109 E/BamHI)
- Make -80 stocks of 228, 230, and 232, 241, 243, and 244
- Make RFP variant of the suicide mechanism to troubleshoot - start with transforming Bca1146 because there isn't enough miniprep to do a digest (actually Chris is doing this for you)
- Make DH10B competent cells
Samanthaliang 13:52, 9 July 2007 (EDT)
- grow up a DH10B culture for competent cells
- Pooled the 228, 230, and 232, 241, 243, and 244
- Note - I pooled 228, 230, and 232 even though they only had 2-5 colonies because I'm getting desperate, will sequence it.
- The spotted plate assay on plates with and without arabinose came out negative for what I wanted. I selected for colonies that lived on Carb plates, and all of them survived on the arabinose plates. It seems in theory that these cells should die, so potentially there could be a problem with it just not triggering the promoter. So now I'm going to grow them up in liquid media to see if that triggers it and then run a growth curve measurement assay with arabinose on the Tecan
here are the steps:
grow up cultures in LB-antibiotic in 96 well blocks
the next day, aim for OD of 0.05 in Tecan plate (about 3ul of overnight into 197ul of media)
add arabinose into the media in order to trigger it
run Tecan growth curve assay upstairs
- I'm pretty scared now that this isn't going to work, so now I really need to push the ceaB variants because maybe those are the ones that are going to work. However, they might potentially be too toxic, since nothing seems to grow on those plates
Samanthaliang 16:04, 7 July 2007 (EDT)
- Water Chris' plants while he's away
- 2AM - Grew up cultures in 96 wells
- Now - Spot on plates with and without arabinose
Samanthaliang 13:38, 6 July 2007 (EDT)
- Miniprep and transform the promoter+suicide cassettes
- Make -80s of cultures
- Religated and transformed 228, 230, and 232 (ceaB variants)
- Religated and transformed 241, 243, and 243 (barnase variants)
- Streaked a new DH10B plate for competent cells
Note - The test replica plates came out pretty badly so I'm not going to replica plate. It's especially difficult because I can't control the cell densities on the plate. Instead, I'm going to grow up cultures in 96 well blocks (maybe 8 rows of each), then dilute, then spot on plates with or without arabinose with the transferpette (the huge multipipetter). Unfortunately because John and I decided it would be safer to pool and miniprep my plates yesterday just in case replica plating went back, now i'm set back a day because I won't have plates to pick colonies from until tomorrow.
Samanthaliang 18:15, 5 July 2007 (EDT)
- Couldn't pool 228, 230 and 232 (the CamR ceaB variants) because nothing grew on those plates. potentially too toxic? Or maybe the digest or ligation just went wrong
- No one seems to have the velvet or stamp for replica plating so I just went to joann's to buy some velvet yesterday but it seems that it may be TOO furry. Might try using the back side of the velvet. Doing some test replica plating to see what works and what doesn't, will also try so nitrocellulose paper (used for Western plots - can't touch or else proteins from hands will stick to it) but that stuff is expensive
- Pooled the promoter+suicide cassettes just in case replica plating goes really badly. Can miniprep them tomorrow and retransform them. Then replica plate the day after tomorrow.
- Made TSS solution
- Poured plates because Arthur left early and almost let the the LB-Carb plates run out
Note - So all the promoter+suicidal cassette subcloning plates came out with colonies. 405 and 413 had the smallest number of colonies although there's no correlation between the two. So this could just be a ligation fluke or something. The ones that showed the most colonies were 407, 408 and 409, which were the ones with barnase. 414, 415, 401, and 402 were okay but had less colonies that all the rest.
Samanthaliang 16:39, 3 July 2007 (EDT)
- Miniprep the suicidal bacteria pools
- Replace CamR with a pBad promoter (pBca9145-Bca1109 - EcoRI/BamHI) to make parts I716401 through I716415
- The sequencing came back good for 217, but 216 had a point mutation unfortunately
- Make -80 stocks of 217 clone and all the suicidal CamR bacteria
- subcloned 228, 230, and 232 using the correct 217, the ceaB variants of suicidal bacteria
- Water Chris' plants while he's away
Samanthaliang 15:12, 2 July 2007 (EDT)
- Austin grew up 216 and 217 for me over the weekend and Nhu miniprepped them
- Analytic digest on 216 and 217 today
- Send clones 216 and 217 that passed analytic digest out for sequencing
- Out of the 18 of the 24 suicidal bacteria - 15 of the plates had enough colonies to pool.
Note: The colonies on these plates were all pretty small, which could indicate that there is some background transcription that is causing them to be slightly toxic even though there is no promoter inserted yet, and there is only a CamR between the EcoRI and BglII sites. Also, the plates that had the fewest colonies (to the extent that I couldn't pool some of them) were the plates that had barnase. Apparently barnase is very hot so that explains why I'm having more trouble getting colonies. Luckily, barnase is an RNA destroyer and may not be as effective in our goal. Apparently I only have to get one of these suicide mechanisms to work, and the DNA destroyers ceaB, BamHI, and BglII probably would be more efficient than the barnase. However, it would be cool if I could get both a RNA and a DNA destroyer into the system. But for now, just worry about the barnases if the rest don't work.
Samanthaliang 18:14, 29 June 2007 (EDT)
- Finished subcloning 216 and 217 by digesting (add 12ul buffer and 1ul of each - DpnI, BglII, BsaI), the clean again elute into 10ul, ligating and transform - ask someone to put them in the fridge
- Finished subcloning 18 out of 24 of the kill mechanisms by ligating and transforming - ask someone to put them in the fridge for me.
Samanthaliang 17:41, 28 June 2007 (EDT)
- Miniprepped everything I grew up - cassette-toxins and rbs-Cre's
- Finished rebuilding 216 and 217 using the dbbs method - unfortunately, the PCR of the ceaB part that I ran yesterday didn't show any bands on the gel by the time Austin cleaned it, I digested it and ran out on a gel. Have to rerun the PCR of 209, the clean it. Can finish it tomorrow.
- Was going to build 18 (24 minus 6 because lack of I716216 and I716217) new biobricks with the 123 method but then I realized that 123 would not work since one of my parts is using a library of rbs. So bascially I've wasted my time with all the transformations. Oh well.
- Going to have to build the whole "kill mechanism" by regular prefix insertion. Made the construction files today. Digested and Gel Purified. Can finish it tomorrow.
- Note: custom ties $15 each iPrint.com
Samanthaliang 14:28, 27 June 2007 (EDT)
- Grew up all the cassette+toxins that have been transformed into Righty
- Grew up the the rbs+Cre variants that have been transformed into Lefty
- Sequencing didn't look good for 216 and 217 clones but looked good for the rbs+Cre pools
- Note - the Ape files I716224, I716225, I716226 are a little weird - I'm not too sure about the pBca1121 sequences that Arthur sent me because they have a pTET promoter between the BglII and BamHI sites instead of a ribosome binding site, but since it would have been a library pool anyway, it didn't matter when I was checking the sequencing because I found the junction sites and made sure that Cre was there, and CamR was definitely there as well so it is safe to assume that the rbs between them didn't just fall out. I think the insert sequence that I need to obtain to make the Ape files perfect would be pBca1121-Bca9230 instead of just pBca1121 - will ask around for this.
- Update - Chris just sent me the file pBca1121-Bca9234, which is a different pool of rbs's but has the same map. So I've updated the ape files
- Made -80 stocks and Clone saved the rbs+Cre pools (keeping the minus 80s with in my old riboregulator pink box) - I feel a little safer doing this because apparently there are some iffy reviews on clone saver cards that say that it loses efficiency quickly.
- Make ape files and construction files for the next round of subcloning which will be
[CamR][rbs][Cre][rbs][ATG][lox][terminator][lox][toxin]
- Colony PCR of 216 and 217
- Lanes 1 through 4 are I716216
- Lanes 5 through 8 are I716217
- Lanes 1 through 4 are I716216
- All of these look wrong and are about 1000 bp short of what the actual product should be...they look like the other gels, where the ceaB was missing. Since this is my 2nd attempt at this one with 123 method and 4th try overall, I think I'm just going to proceed with the rest of the toxins and then subclone this one later. Chris thinks that it's possible that it may be toxic. Will try to subclone using the dbbs method and attach rbs+Cre to it later. Right now, I want to get moving with the rest of the toxins though because I want to do some assays soon and see some results.
- Set up PCR for dbbs method 216 and 217
Samanthaliang 12:59, 26 June 2007 (EDT)
- Miniprep the I716217 clones and sequence them
- Miniprep I716216 clone that passed colony PCR test and sequence it
- Miniprep the rbs+Cre variants (I716224, I716225, I716226) and send them out for sequencing
- Just in case, tried a 123 method again for 216 and 217 because I'm not entirely confident from the gels from the colony PCRs.
- Make construction files for rbs+Cre+cassette+toxin variants. I think I'm just going to 123 them actually.
- Transform all cassette+toxins into the Righty (exceptions are 217 and 216 which are already in that strain) and all rbs+Cre variants into Righty
Samanthaliang 17:34, 25 June 2007 (EDT)
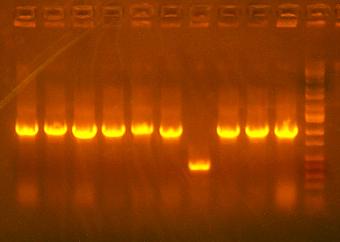
- Colony PCR on I716216, then maybe grow them up at the end of the day
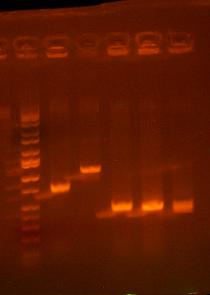
ladder, clone 1, clone 3, clone 4, clone 5, clone6
all these bands look too small...should be about 2195 bp. but will grow up and sequence clone 3 just in case.
- Scrape rbs+Cre variant plates - make sure you pool them because you have a library of rbs's
- Grow up clones 4 and 7 of I716217 from the colony PCR plate
Note: Chris has updated the 123 method of subcloning composite parts so that it has about 90% correct yield now. I should transform all of my toxins into Righty once they're ready. The ceaB variants should already be in them. I will do that once it has been confirmed that the rbs+Cre variants are correct, and transform those in parallel into Lefty. Then it should be super easy! No gels or maps necessary!
- Showed high schoolers how to poor plates
Samanthaliang 14:42, 22 June 2007 (EDT)
THE LAB FLOODED!
- Checked sequencing for cassette toxins and Richard likes the cartoon e.coli that I draw on the envelopes
- Got a good clone of every cassette toxin except for the ceaB variants. Good thing I tried the 1-2-3 method on those.
- 216 - still working on it, can 123 it
- 217 - have a colony PCR plate of it that should be right
- 218 - clone 2
- 219 - clone 1
- 220 - clone 1
- 221 - clone 1
- 222 - clone 3
- 223 - clone 4
- Do the 123 method on I716216
- Move equipment from wall benches to bays because they're going to be fixing the water damage
Samanthaliang 14:22, 21 June 2007 (EDT)
- Miniprep the cassette toxins, send them out for sequencing, clone save
- Subclone the rbs+Cre variants
- Colony PCR for the I716217 from the 1-2-3 method
Samanthaliang 18:48, 20 June 2007 (EDT)
- Miniprep Lefty and Righty strains
- Tried the 123 method for I716217 even though I may have already gotten that part via regular subcloning. Plated the product of it today and will probably have to do a colony PCR tomorrow
- Checked sequencing on the GTG Cre and it finally turned out right! third time's the charm i guess. Anyway, so from now on, clone 2 of I716212 is the correct one to use
- I clone saved the 212 clone that was correct and also updated the spreadsheets to show which ones had clones that failed the sequencing test.
- I tried to subclone the rbs libraries with the Cre variants but totally failed. I kept messing up with elutions while cleaning it up. Anyway, I'll try again tomorrow when I'm a little more focused, probably with the Cam variant of it. But I need to make ones with all 3 Cre variants (ATG, GTG, and TTG start). The construction files for these parts are already made.
- Grew up the following cultures of the cassette toxins from the colony PCR plates
- 216-1, 216-3
- 217-1, 217-2
- 218-1, 218-2
- 219-1, 219-2
- 222-1, 222-3
- 223-1, 223-4
Samanthaliang 15:26, 19 June 2007 (EDT)
- Grow up one colony of 214 and 215 in Lefty, and 210 and 209 in Righty
- mini 212 and sequence it again
- actually didn't mini the 223s and do an analytic gel to see if any are good because I did a colony PCR and they came out promising
- Plates of the new cassette+toxins looked great
- Colony PCR on the cassette+toxins with ca998 and g01001
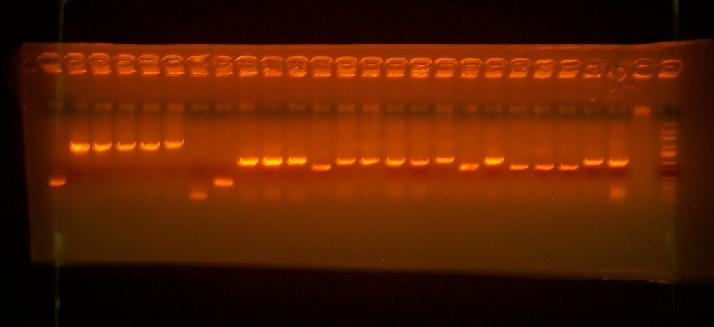
216-1, 216-2, 216-3, 216-4, 217-1, 217-2, 217-3, 217-4, 218-1, 218-2, 218-3, 218-4, 219-1, 219-2, 219-3, 219-4, 222-1, 222-2, 222-3, 223-1, 223-2, 223-3, 223-4, 2223-5, blank, ladder
Looks like there's promising clones for each construct. Can grow them up tomorrow.
Samanthaliang 14:54, 18 June 2007 (EDT)
- grew up 2 clones of 212 that i subcloned again
- sequencing came back for the cassette+toxins and only 220, 221, 222 are good.
- Subcloning all the others again with more DNA in the digest.
- Also, will grow up some more colonies of 223 to screen tomorrow because it uses the same insert as 222 and the same backbones as 220, 221, so it is possible that some of the colonies may be correct.
- Also, will try out the 123 cloning method on 216 and 217 (218 and 219 too). First step is to transform the inserts and backbones parent plasmids into the special strains. Transformed 214 and 215 into "Lefty" competent cells, then 209 and 210 into "Righty" competent cells.
Samanthaliang 18:10, 15 June 2007 (EDT)
- miniprep 216-223 clones
- test digest 216-223 clones
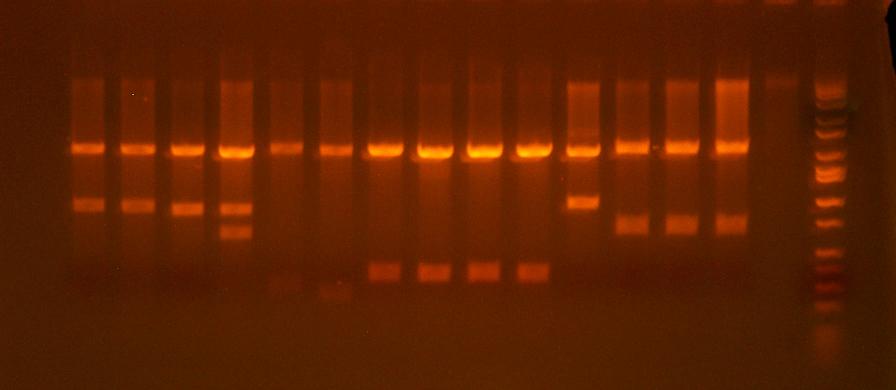
from left to right:
I716216-1, I716216-2, I716217-1, I716218-2, I716218, I716219,I716220-1, I716220-2, I716221-1, I716222-1, I716222-2, I716223-1, I716223-2, blank, ladder
- clones that pass the test digest: 220-1, 220-2, 221-1, 221-2, 222-1
- I'm also clone saving and sequencing 216-1 and 223-1 just because I want to see what's up with them. 216 and 217s don't show the pattern of either the desired product or either vector. 223-1 is a maybe, it may be missing the cassette. 218 and 219 both failed miserably, but that was to be expected.
- accidentally grew up 212, 218 and 219 on plain LB plates with no Carb. Arg. Religate and transform again from previous digests
- If the 218 and 219 plates come out poorly again, will have to redo the I716210 digest (BglII/BglI), should have enough of the cassette though.
- Read that RNA destabilization article
Samanthaliang 15:28, 14 June 2007 (EDT)
- sequencing for the GTG start Cre was same as before - still has an ATG start - I can't really figure out what is going wrong here. I definitely used dpnI to digest the parent vector because I remember having to go upstairs to the Keasling lab to get hooked up with that enzyme since we were out of it downstairs. Also, both times I did it, there were a lot of colonies on the plate, and there would have been none if it were the parent vector since I used dpnI. It must be some problem with the PCR then - is it self-correcting? I double checked the oligo and it definitely has a GTG start. I guess I'll try to make it one more time, but if it doesn't work this time then I might have to try Quick Change if it's worth having this part.
- Grow up colonies for cassettes+toxins (216-223)- the plates are alright. except for the ones with BamHI, those plates only had one colony each. weird. maybe a problem with the digest since both of them are bad? Will re-ligate and transform today hoping for an easy fix. If the sequencing comes out bad though eventually, I will have to redo the subcloning for 218 and 219 from the start.
- Make the ape files for cassette+toxins
- Read that locks and keys article
- Subclone Cre (GTG) over.
Samanthaliang 18:21, 13 June 2007 (EDT)
- miniprepped 212s - sequenced the clones that came from the plate with more colonies (put cultures in fridge)
- subcloned all the cassettes+toxins (216-223) because sequencing came back good on the cassettes for the clone 2s - 8 different new plasmids!
- put teeth on the centrifuge
Samanthaliang 15:26, 12 June 2007 (EDT)
- grew up 212 clones
- miniprepped 214 and 215 clones and sent them out for sequencing
- clone saved
- sequencing came in and it looks like we have a good clone for every toxin!
- ceaB (209) - clone 2
- BamHI (210)- clone 2
- barnase (211)- clone 1
- BglII - already a miniprep Bca1147
- still missing Cre with a GTG start - is being grown up today
- Cre with a TTG start (213) clone 2 is perfect
- Made construction files for cassette+toxin
- Now in charge of the google spreadsheets - have to invite people and what not
For tomorrow: Once the cassettes have been confirmed for sure, can digest those and the toxins and ligate.
Samanthaliang 19:24, 11 June 2007 (EDT)
- grew up 214 and 215 clones
- Sequenced 209 (clone 1&2), 211 (clone 1&2), and 213 (clone 2) again with G01001 (backwards) because the genes were too big to confirm for sure that they were right, 210 was perfect the first time
- Redid the PCR for 212 (Cre-GTG) because both sequences came back with the ATG start codon still
- Subcloned 212
- Clone saved and recorded on the new Google Spreadsheet
- emailed china team that wanted our oriT knockouts from last year - told them to look in the registry again
Samanthaliang 20:00, 9 June 2007 (EDT)
- Miniprepped 209, 210, 211, 212, 213 and sent them out for sequencing
- Sequencing came in for 207 and 208 - should use clone 1 for both.
Will try to come in on Sunday too to grow up the 214 and 215 cultures - i moved the plates to the fridge for now.
Samanthaliang 17:58, 8 June 2007 (EDT)
- Made a ridiculously large batch of DH10B competent cells
- Miniprepped 207 and 208 clones, clone saved them, and sent them out for sequencing
- Used those preps to subclone I716214 and I716215 (two versions of each)
- Grew up cultures of 209, 210, 211, 212, 213
Samanthaliang 17:58, 7 June 2007 (EDT)
- Chris gave me the wrong template for Cre and I didn't notice until today so I redid those 2 PCRs
- All the other PCRs looked fine on a gel
- Cleaned up Digested and inserted all PCR products into Biobrick form (I716210 through I716113)
- Got sequencing data back and only one of the [rbs][atg] ones came out good, but the [lox][terms]s all came out good - just forget about the [rbs][atg] ones for now. More detail - the ones that failed also had really tiny colonies and were in Bca1101 form with a promoter in front, so it might be translating some nonsense that is toxic to the e.coli - but since I have one of the rbs's already I'm just going to proceed with that
- Picked colonies from pBca9145-I716207-1 and pBca9145-I716208-1
- Austin grew up 10ml of DH10B for me so I can make competent cells tomorrow
Samanthaliang 17:31, 6 June 2007 (EDT)
- miniprepped 201 through 206 and sent them out for sequencing
- put the clones on "clone saver" paper
- subcloned 207 and 208 with all clones (have 2 versions of each just in case)
- PCRed ceaB, BamHI, Barnase, Cre with both alternate stop codons
Samanthaliang 19:20, 5 June 2007 (EDT)
- Grew up 2 colonies each of I716201 through I716206
- Made a lot of construction files and entered things into the registry
- My oligos were ordered today!
- Demonstrated liquid media making and registry entering
- Now will also make an RFP variant as a reporter on how well Cre is working
To do tomorrow:
- subclone 207 and 208
- Run PCRs if they come in the afternoon
- Send things out for sequencing and make -80s
- Fill out and sent those survey forms
Samanthaliang 15:29, 4 June 2007 (EDT)
- Oligos to make biobricks of toxins are not yet in - maybe Wednesday or Thursday?
For cassette between Cre and toxin: [rbs][ATG][Lox][Term][Lox]
- Made [rbs][ATG] variants with different ribosome binding sites
I716201 is Bca1106A.Bca1112
I716202 is Bca1106B.Bca1112
I716203 is Bca1128A.Bca1112
I716204 is Bca1090.Bca1112
- Made [Lox][Term] variants with 2 different terminators (will probably use Bca1092 version)
I716205 is Bca1114.Bca1124
I716206 is Bca1114.Bca1092
- Tomorrow will make [Lox][Term][Lox] part
- Then will put the rbsATG together with LoxTermLox
eek so many construction files and ape files to make.
to do