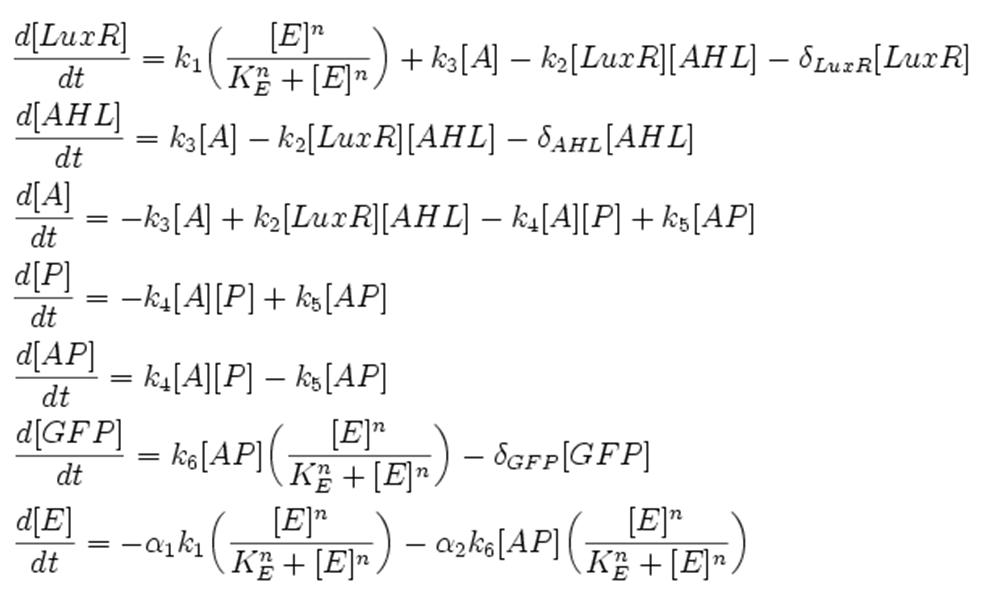Imperial/Infector Detector/Modelling
From 2007.igem.org
(→Introduction) |
|||
| Line 21: | Line 21: | ||
==Introduction == | ==Introduction == | ||
Infector Detector (ID) is a simple biological detector, which serves to expose bacterial biofilm. It functions by exploiting the inherent AHL (Acetyl Homoserine Lactone) production employed by certain types of quorum-sensing bacteria, in the formation of such structures.<br> | Infector Detector (ID) is a simple biological detector, which serves to expose bacterial biofilm. It functions by exploiting the inherent AHL (Acetyl Homoserine Lactone) production employed by certain types of quorum-sensing bacteria, in the formation of such structures.<br> | ||
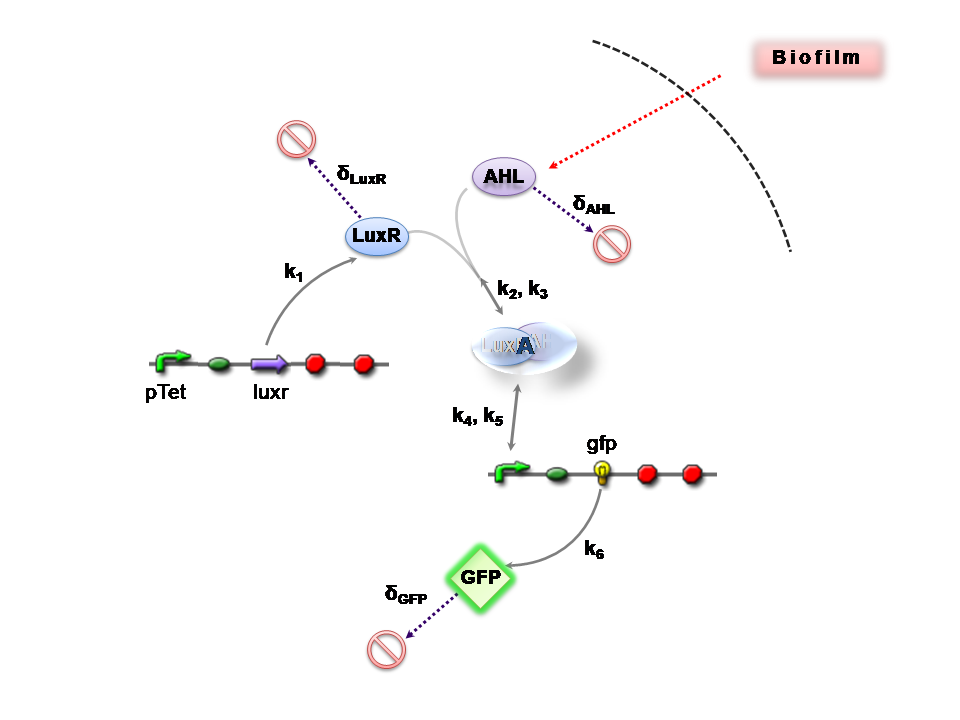
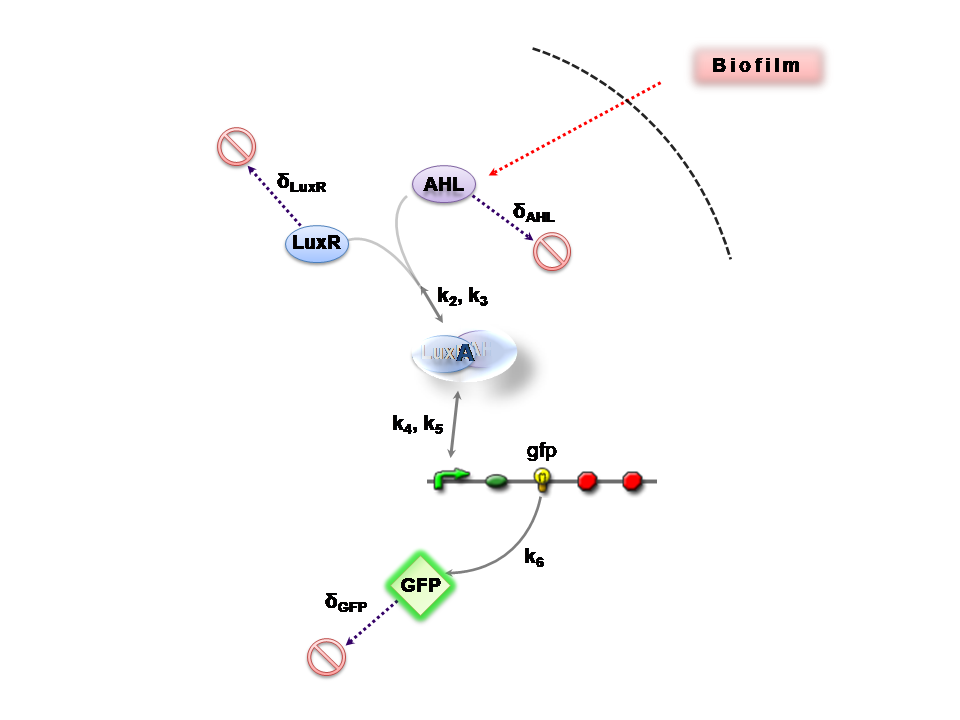
| + | This section presents a preliminary model for an AHL detector, which employs the backbone of the Lux quorum sensing feedback mechanism. Figure 1 illustrates the full system we are investigating. | ||
In the design phase, two possible system constructs were proposed, as a solution to the problem of detecting AHL-producing biofilm. | In the design phase, two possible system constructs were proposed, as a solution to the problem of detecting AHL-producing biofilm. | ||
| + | According to our specifications, the most crucial feature of this system is the sensitivity to a minimum [AHL] of 5nm. In other words, this represents the minimal AHL concentration for appreciable expression of a chosen reporter protein. | ||
| + | Furthermore, we attempt to establish a functional range for possible AHL detection. How does increased AHL concentration impact on the maximal output of reporter protein? | ||
| + | Finally, how can the system performance be tailored, by exploiting possible inputs to the system (e.g. varying initial LuxR concentration and/or concentration of pLux promoters). | ||
| + | How does such customization impact upon the maximal output of fluorescent reporter protein and/or response time? | ||
| - | + | We attempt to answer these questions by conducting simulation of the system ''in-silico''. | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
==Implementation & Reaction Network== | ==Implementation & Reaction Network== | ||
Revision as of 00:34, 24 October 2007

Introduction
Infector Detector (ID) is a simple biological detector, which serves to expose bacterial biofilm. It functions by exploiting the inherent AHL (Acetyl Homoserine Lactone) production employed by certain types of quorum-sensing bacteria, in the formation of such structures.
This section presents a preliminary model for an AHL detector, which employs the backbone of the Lux quorum sensing feedback mechanism. Figure 1 illustrates the full system we are investigating.
In the design phase, two possible system constructs were proposed, as a solution to the problem of detecting AHL-producing biofilm. According to our specifications, the most crucial feature of this system is the sensitivity to a minimum [AHL] of 5nm. In other words, this represents the minimal AHL concentration for appreciable expression of a chosen reporter protein. Furthermore, we attempt to establish a functional range for possible AHL detection. How does increased AHL concentration impact on the maximal output of reporter protein? Finally, how can the system performance be tailored, by exploiting possible inputs to the system (e.g. varying initial LuxR concentration and/or concentration of pLux promoters). How does such customization impact upon the maximal output of fluorescent reporter protein and/or response time?
We attempt to answer these questions by conducting simulation of the system in-silico.