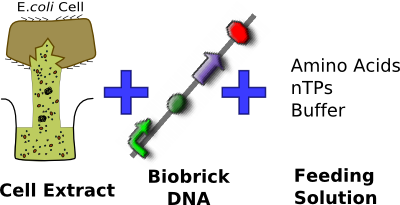Imperial/Infector Detector/Design
From 2007.igem.org

Infector Detector: Design
Design Overview
| Specification | Design Solution | System Level |
| Health & Safety - System must not contain living cells, or cause any harm to patient. | Use a Cell Free System e.g. Promega's S30 Cell Extract | Chassis |
| Shelf-life - System must have a shelf life of 7 days. | Use a Cell Free System that can be packaged and stored in a -80°C freezer for 7 days. | Chassis |
| Application - System must be applied as a cream or spray. | Package the system in liquid form. | Chassis |
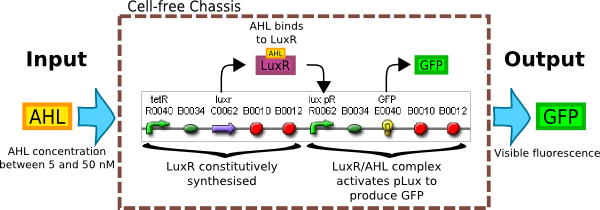
| Inputs - System must be sensitive to AHL concentration between 5-50nM. | Sensitive AHL receiver to detect low AHL levels - Part no. F2620. | Construct |
| Outputs - System must give a visual signal if AHL is present. | Couple AHL to a reporter system expressing fluoresent protein eg. RFP | Construct |
| Operating Conditions - System must operate within temperature 20°-30°C. | Gene expression systems and protein must be thermostable. Characterise a Cell Free System at these temperatures. | Construct & Chassis |
| Response Time - System needs to have a response time under 3 hours. | To be Determined - this is hard to design for. Protease Inhibitor of Cell Extract should ensure degradation of Visual Reporter is minimal. | Construct & Chassis |
DNA Constructs
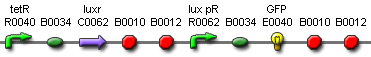
Our aim was to create a system that can quickly detect a small (5nM) concentration of AHL in solution. We needed a well characterized detector system sensitive to 3OC6HSL which we will attach to a standard GFP reporter to determine its response to GFP. The first construct is appealing as the AHL receiver in front is well-documented as part [http://partsregistry.org/Part:BBa_F2620 BBa_F2620], thus giving more assurance in the reproducible nature of our results.
However, the weakness of construct 1 lies in the fact that it does not respond uniformly to constant AHL concentration since the activator protein LuxR is produced by a promoter and not maintained at a constant level. This makes it hard to relate the output of the system to the AHL input. However, if purified LuxR could be added to the system instead, the construct could be simplified by removing the constitutive LuxR production. This would give us more control of the input, and therefore better reproducibility.
Chassis Selection
We have chosen to use the commercially available S30 E. coli cell extract made by Promega. After looking into a variety of different cell-free systems, we decided that this chassis is most suitable. In particular, it allows us to comply with the Health and Safety regulations of the field we are working in, as we do not want replicative bacteria that could potentially be pathogenic to come in contact with urinary catheters.
In addition to complying with health regulations, the S30 cell extract is commercially available, meaning that it has been shown to work. (1) This allows our focus to be on tuning the chassis to suit our needs, rather than trying to make the chassis work in the first place.